Trends in Chemotherapy Patterns and Survival of Patients with Advanced Gastric Cancer over a 16-Year Period: Impact of Anti-HER2–Targeted Agent in the Real-World Setting
Article information
Abstract
Purpose
This study aimed to evaluate the survivals of patients with metastatic or recurrent gastric cancer (MRGC) over a period of 16 years and to investigate the recent changes in chemotherapy patterns.
Materials and Methods
A total of 5,384 patients who received chemotherapy for MRGC between 2000 and 2015 were analyzed. The analysis focused on a comparison of the first-line chemotherapy between four periods: 2000–2003 (period 1), 2004–2007 (period 2), 2008–2011 (period 3), and 2012–2015 (period 4).
Results
There were 880 patients (16%) in period 1, 1,573 (29%) in period 2, 1,435 (27%) in period 3, and 1,496 (28%) in period 4. Cytotoxic doublet-based therapy was the most commonly used (78%) first-line chemotherapy, and the combination of trastuzumab and doublet chemotherapy was provided to 288 patients. The overall survival (OS) rates at 12 and 24 months were steadily improved as follows: 39.2% and 14.6% in period 1, 43.5% and 17.6% in period 2, 50.3% and 20.6% in period 3, and 51.7% and 24.1% in period 4, respectively (p < 0.001). Among the patients who received the doublet-based chemotherapy, the median OS of those who received trastuzumab was 18.0 months (95% confidence interval [CI], 15.5 to 20.6), while that of those who received other doublet therapies was 11.2 months (95% CI, 10.8 to 11.6).
Conclusion
The OS was improved over time with advancements in chemotherapy, particularly the introduction of the anti-HER2–targeted agent, which contributed to the increase in the number of long-term survivors and established the superiority of OS for the treatment of MRGC.
Introduction
Gastric cancer is one of the most common causes of cancer-related mortality worldwide [1]. In Korea, gastric cancer is the second leading malignancy in terms of incidence rate and the fourth leading cause of cancer-related deaths [2]. At present, systemic therapy is considered as the primary treatment in patients with advanced unresectable or non-curatively resected gastric cancer [1,3]. Nonetheless, the median survival of patients with metastatic or recurrent gastric cancer (MRGC) is approximately 1 year in clinical practice.
Over recent years, with advancements in the pathophysiological understanding, other tumors, such as lung, or colorectal cancers, have increased the biomarker-driven treatment options leading to individual treatment plans and improvements in the 5-year overall survival (OS) rate from less than 5% to 25%–40% in patients with advanced or metastatic disease [4,5].
Until the 2000s, most clinical trials for patients with MRGC were focused on the introduction of new cytotoxic agents or novel combinations of agents or on the modification of existing regimens [6–8]. However, since the 2010s, the trend of clinical trials was changed to actively explore the biomarker-driven targeted agents or novel agents in combination with cytotoxic chemotherapy [9–13]. The most representative example of this is trastuzumab, an anti-human epidermal growth factor receptor 2 (HER2)–targeted agent, which is now a standard agent used in patients with HER2-positive gastric cancer [9]. Similarly, ramucirumab, a monoclonal anti–vascular endothelial growth factor receptor 2 antibody, demonstrated prolonged survival in advanced gastric cancer patients in a second-line setting [14,15].
Recently, real-world analysis is increasingly being used because data from these analyses can be utilized for comparable validations of clinical trials [16]. Furthermore, the clinical trial data may not be representative of the general population and may underestimate clinically essential subgroups in real practice.
Previously, we reported an increase in the survival rates of patients with MRGC over a period of 12 years due to advances in chemotherapy and supportive care [17]. This study aimed to update the survival outcomes of the patients using a prospectively collected registry and to investigate the causes for the change in survival over a period of 16 years.
Materials and Methods
1. Patients and data collection
All patients who were treated for advanced gastric cancer between January 2000 and December 2015 at the Asan Medical Center (Seoul, Korea) were retrospectively screened. A total of 5,931 patients who received first-line palliative chemotherapy for MRGC were identified. Patients who were ≥ 19 years of age with histologically confirmed adenocarcinoma of the stomach and who had received at least one cycle of chemotherapy were included in the study. Those who had a history of other malignancies, an unknown start date of first-line chemotherapy at other hospitals, or microscopic residual tumors after prior R1 resection were excluded. Of the 5,931 patients screened, 5,384 fulfilled the inclusion criteria. The records of the patients, stored in a prospectively collected registry, were reviewed for information regarding the demographic data, tumor characteristics, type of treatment, treatment responses, and survival. The last update for each patient was obtained in October 2018 until the date of death or the end of the follow-up period. The analysis focused on four intervals: 2000–2003 (period 1), 2004–2007 (period 2), 2008–2011 (period 3), and 2012–2015 (period 4).
Chemotherapy was categorized according to the regimen type as follows:
single (fluoropyrimidine [F], taxane [T] or irinotecan [I]);
doublet (F/platinum [P], T/P, F/I, F/T, T/I, anthracycline [A]/P, or A/F);
doublet/Tmab (F/P with trastuzumab);
doublet/novel (F/P with novel agents in clinical trials);
triplet (F/P/T);
single/Rmab (T with ramucirumab);
immunotherapy (immune checkpoint inhibitors).
The patients were grouped according to our prognostic model for MRGC [18]. This model used a scoring system consisting of eight prognostic factors: Eastern Cooperative Oncology Group (ECOG) performance status (PS) of ≥ 2 or bone metastasis (2 points each); absence of gastrectomy, peritoneal metastasis, or lung metastasis; alkaline phosphatase level > 120 IU/L; albumin level < 3.3 g/dL; and total bilirubin level > 1.2 mg/dL (1 point each). The patients were divided into good (0–1 point), moderate (2–3), and poor (≥ 4) risk groups based on the scores.
2. Statistical analysis
Differences in patient and tumor characteristics between the four periods were analyzed using the chi-square test or ANOVA test. OS was measured from the date of starting the first-line chemotherapy until death from any cause. Progression-free survival (PFS) was measured from the date of starting the first-line chemotherapy until tumor progression or death from any cause. The survival rates were estimated using the Kaplan-Meier method and compared using the log-rank test. Prognostic factors were analyzed using Cox proportional hazard regression models. A two-sided p-value < 0.05 was considered significant, and 95% confidence intervals (CIs) were calculated. Using a conservative Bonferroni correction for multiple comparisons, type I errors were adjusted. All statistical analyses were performed using the SPSS ver. 25.0 software package (IBM SPSS Statistics, Armonk, NY).
Results
1. Baseline characteristics over a 16-year period
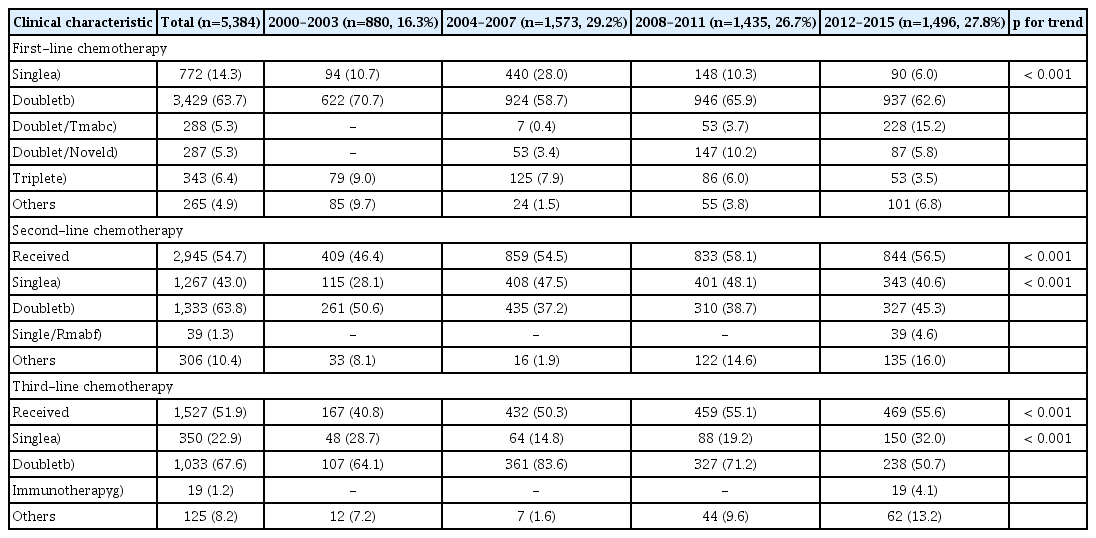
Between 2000 and 2015, 5,384 patients received palliative chemotherapy as first-line treatment for MRGC: 880 (16%) in period 1, 1,573 (29%) in period 2, 1,435 (27%) in period 3, and 1,496 (28%) in period 4. The median age of the patients was 56 years (range, 19 to 91 years), and about 36% of the patients presented with recurrent gastric cancer over the 16 years. The clinical characteristics of the patients are shown in Table 1. Slight but significant changes in the following characteristics were observed between period 1 and the other three periods: ≥ 65 years age; poor ECOG PS (2/3); two or more metastases; peritoneal, lung, or bone metastasis; and poor-risk group in the prognostic model over time (p for trend < 0.001).
2. Treatment characteristics over the four periods
In the first-line chemotherapy, cytotoxic doublet-based treatment was most commonly used (74%) followed by a single-agent (14%). The results of the analysis of the chemotherapy data during the four periods are summarized in Table 2. About 11% of the patients (n=581) were treated using new agents, and the use of the trastuzumab-combined doublet therapy was increased up to 15% during period 4 when compared to the other three periods (4%). Second-line chemotherapy was administered to 55% of the patients, and 52% of them received the third-line chemotherapy. The proportion of patients who received the second- and third-line treatments was significantly increased over periods 2, 3, and 4 when compared to period 1 (p < 0.001). In period 4, the ramucirumab-combination chemotherapy and immunotherapy were introduced, and given to about 4% to 5% patients in the second-line and third-line treatment, respectively.
3. Response rate according to the first-line treatment
Of the 2,525 patients (47%) with measurable disease, about 42% achieved a partial response and 3% (n=66) achieved a complete response, making the overall response rate (ORR) as 44.4% (95% CI, 41.5 to 47.3) (Table 3). According to the first-line treatment, the ORR was higher in patients who received trastuzumab-combined doublet therapy (68%) and triple therapy (59%) compared to those who received other treatments.
4. Survival outcome over the four periods
Overall, 94% of the patients (n=5,060) died, with a median OS of 11.0 months (95% CI, 10.7 to 11.4). The median follow-up time for the living patients was 74.5 months (interquartile range, 48.0 to 133.4 months). The median OS (95% CI) and long-term survivals steadily improved over the study period: 9.6 months (9.0–10.2) in period 1, 10.3 months (9.7–10.8) in period 2, 11.9 months (11.2–12.6) in period 3, and 12.2 months (11.5–12.9) in period 4 (log-rank, p < 0.001) (Fig. 1A). In contrast to the steady increase in OS curves from periods 1 to 4, the PFS curves were similar in periods 1 (5.6 months, 5.2–6.0), 2 (5.3 months, 5.0–5.7), and 3 (5.8 months, 5.5–6.2), with a significant increase in period 4 (7.0 months, 6.5–7.4; p < 0.001) (Fig. 1B). In terms of the survival rate, the OS (95% CI) at 12 and 24 months demonstrated a steady rise as follows: 39.2% (33.8–44.6) and 14.6% (8.2–21.0), respectively, in period 1; 43.5% (39.8–47.2) and 17.6% (13.1–22.1), respectively, in period 2; 50.3% (46.6–54.0) and 20.6% (15.9–25.3), respectively, in period 3; and 51.7% (48.0–55.4) and 24.1% (19.5–28.7), respectively, in period 4 (p < 0.001) (S1 Fig.).
5. Survival outcome according to the first-line treatment
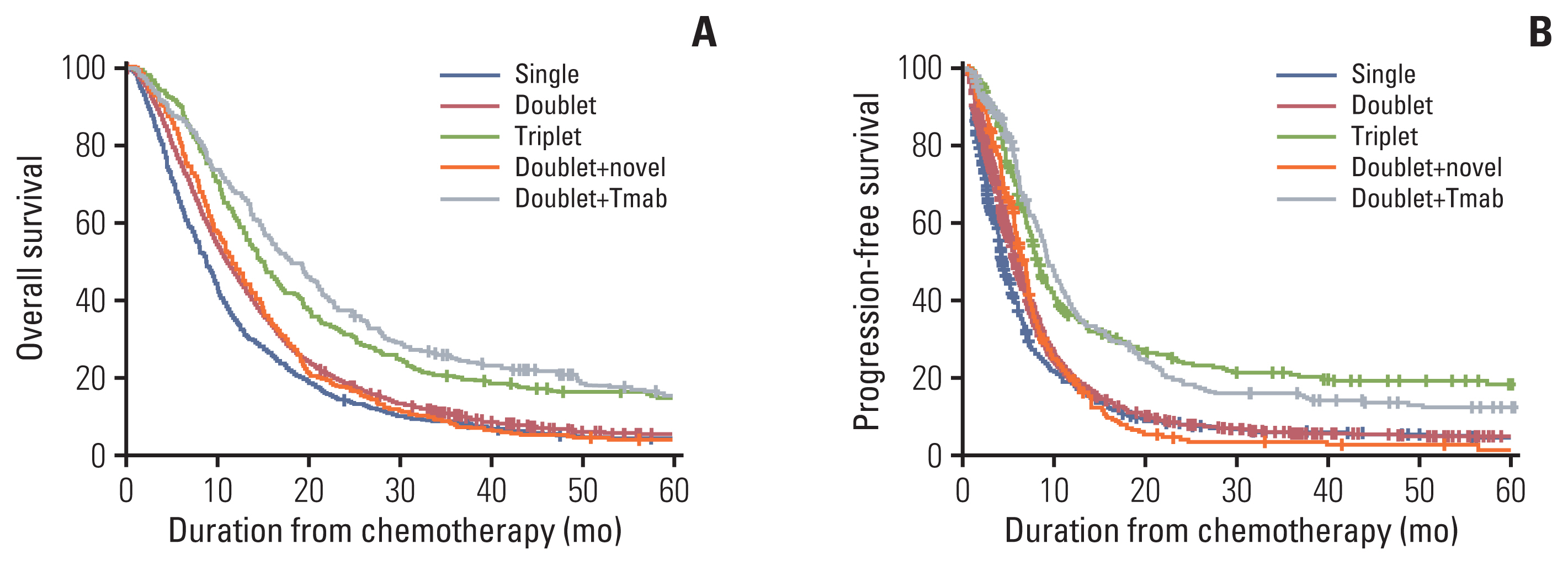
According to the first-line treatment, the median OS was 8.7 months in patients who received the single-agent therapy, 11.0 months in those who received the doublet therapy, 11.7 months in those who received doublet therapy with novel agents, 18.0 months in those who received doublet therapy with trastuzumab, 14.7 months in patients who received triplet therapy, and 10.0 months in the others group (p < 0.001) (Fig. 2A). The median PFS associated with the treatments were as follows: 4.2 months in single-agent, 5.7 months in doublet, 6.7 months in doublet with novel agents, 9.3 months in doublet with trastuzumab, 8.0 months in triplet, and 4.5 months in the others group (p < 0.001) (Fig. 2B). Similar to the results of the treatment response, OS and PFS were longer in patients treated with trastuzumab-combined doublet therapy and triple therapy compared to those of doublet therapy.
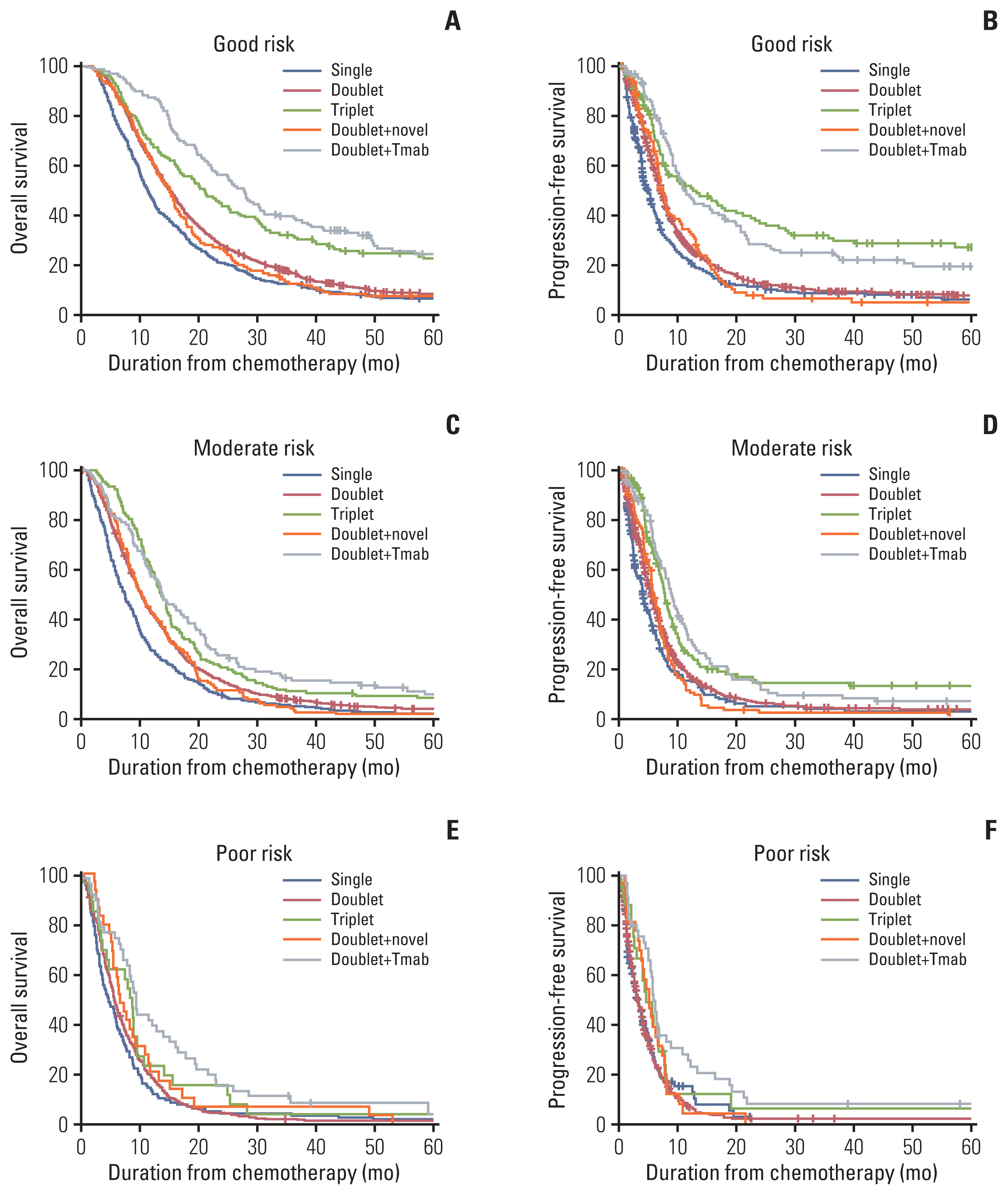
6. Comparison of survival outcomes according to the risk group
To rule out the possible unbalanced distribution of patients in each treatment, the survival outcomes were compared according to the prognostic risk model (Fig. 3, S2 Table). Regardless of the prognostic groups, the patients treated with trastuzumab-combined doublet therapy had better OS and PFS compared to those in the other treatment groups, including those in the poor-risk groups.
Discussion
This study analyzed the trend in the changes in palliative chemotherapy and treatment outcomes of patients with MRGC using real-world data from a single institution over a period of 16 years. Although the median OS of the period 4 was slightly improved compared to the period 3 (12.2 months vs. 11.9 months), it is notable that the long-term survival was steadily improved over the time: the OS rate at 12 and 24 months was improved from 39% to 52% and 15% to 24%, respectively.
1. Survival of trastuzumab plus doublet chemotherapy in real practice
The combination of trastuzumab and doublet chemotherapy was introduced in the ToGA trial since period 2 [9]. Its administration was gradually increased from June 2013 (period 4) when it was reimbursed and eventually expanded to 15% of the patients. In the present study, the antitumor efficacy of the trastuzumab plus doublet chemotherapy was remarkably improved when compared to the doublet chemotherapy without trastuzumab (with or without novel agents). Even after the categorization of the patients according to the prognostic scores, the OS in patients who received a combination of trastuzumab was significantly better than those in patients who received other doublet therapies (p < 0.001).
Notably, the ORR (67% vs. 47%), PFS (9.3 months vs. 6.7 months), and OS (18.0 months vs. 13.8 months) values obtained in the current study were higher than those reported in the ToGA trial [9]. This may be because the patients in this registry received a combination of trastuzumab and doublet therapy when immunohistochemistry (IHC) HER2 3+, or 2+ and fluorescence in situ hybridization (FISH)–positive in contrast to the eligibility of ToGA trial, where the median OS also increased from 13.8 to 16.0 months in the subgroup analysis of the patients with IHC HER2 3+, or 2+ and FISH-positive tumors only [9].
When the patients treated with the trastuzumab plus doublet chemotherapy were excluded, the median OS of the patients in periods 3 and 4 were similar at 11.6 (95% CI, 10.9 to 12.3) and 11.4 months (95% CI, 10.7 to 12.1), respectively. Furthermore, the survival rates at 24 months were similar at 19.2% and 21.4%, respectively. As increasing up to 15% of the combination of trastuzumab plus doublet chemotherapy in the period 4, long-term survivors may be increased. This could imply that the addition of trastuzumab might have been critical for OS improvement over the past 16 years.
2. Single-agent therapy for first-line chemotherapy in real practice
The fluoropyrimidine/platinum doublet-based chemotherapy has been considered as a standard first-line treatment; therefore, it was provided to most of the patients (74%) in this study. However, some patients who received different chemotherapies as first-line therapy presented with distinct clinical features (S3 Table). The patients who were treated by single-agent chemotherapy can be broadly classified into fluoropyrimidine and taxane groups (S4 Table). Fluoropyrimidine single chemotherapy was provided to patients who were > 70 years old (52%) or had poor PS (ECOG 2/3; 20%). Therefore, these patients received less subsequent chemotherapy (second-line; 35%) resulting in a poor prognosis.
3. Single-agent therapy for recurrent disease
Among the 421 patients who received a single-agent for recurrent disease, the number of patients who received fluoropyrimidine (n=203, 48%) and taxane (n=218, 52%) were evenly distributed. Of the 323 patients with available information about the recurrence and prior surgery dates, those who received taxane had a median recurrence-free survival (RFS) of 13.0 months, whereas those who received fluoropyrimidine had a median RFS of 22.4 months. These findings indicate that the patients who received a single taxane agent had recurrence during or soon after the adjuvant chemotherapy with fluoropyrimidine +/− platinum; fluoropyrimidine was given to older patients or to those with poor PS because it would be difficult for them to endure the combination chemotherapy regardless of metastasis or recurrence. In the single taxane group, the tumors are considered resistant to prior chemotherapy with fluoropyrimidine +/− platinum. The median PFS in the single-agent taxane group was inferior to those in single-agent fluoropyrimidine group (3.9 months vs. 4.6 months; hazard ratio [HR], 1.28; 95% CI, 1.07 to 1.53) and in the salvage chemotherapy group in a phase III trial [19]. Furthermore, the OS of the taxane single-agent group was also inferior to that of the fluoropyrimidine single-agent group (8.7 months vs. 10.6 months; HR, 1.43; 95% CI, 1.18 to 1.75), despite similar proportions in the prognostic group and the fact that more patients receive subsequent chemotherapy (S5 Fig.).
4. Triplet chemotherapy as first-line treatment
When triplet chemotherapy is given as a neoadjuvant setting in patients with locally advanced but resectable gastric cancer, the triplet could provide survival improvement by increasing the downstaging and R0 resection rate in the absence of distant metastases, such as the results of FLOT4 or PRODIGY study [20,21]. In contrast, it has not been proven whether the triplet is better than the doublet in the metastatic setting. In locally advanced unresectable or minimally metastatic gastric cancer, a phase II AIO-FLOT3 trial reported that preoperative fluorouracil, leucovorin, oxaliplatin, and docetaxel (FLOT) showed a median survival of 31 months, and further phase III study is ongoing [22]. In our registry, about 30% of the patients had an initially unresectable but locally advanced disease, and a considerable number of patients (n=90) could undergo curative-intent surgery after triplet chemotherapy and had a median survival of 33.2 months (95% CI, 21.7 to 44.8). Therefore, the triplet chemotherapy can be considered, particularly when the PS of the patients is excellent and sequential curative-intent surgery may be possible.
5. Additional reasons for improvements in survival outcomes
There are several other explanations for improvements in treatment outcomes over the years. The proportion of patients who received sequential second- or third-line chemotherapy increased. Two phase III trials demonstrated the benefits of second-line or additional chemotherapies for the improvement of the survival outcome in patients with advanced gastric cancer [19,23]. Therefore, second-line systemic therapy is established as a standard treatment if the patient’s PS and major organ functions are preserved [3]. Among the targeted therapies, trastuzumab, ramucirumab, and, recently, the immune checkpoint inhibitor nivolumab have been shown to be effective against advanced gastric cancer [14,15,24]. However, it is difficult to analyze the efficacy and survival outcome of the ramucirumab-combination chemotherapy or immunotherapy in this study population because they were introduced in period 4. Therefore, the current analysis focused on the efficacy and impact of trastuzumab on the treatment outcomes.
In summary, this analysis presents the trends in clinical chemotherapy patterns and treatment outcomes in patients with MGRC over the past 16 years using real-world data. The OS was improved over time with advancements in chemotherapy, particularly the introduction of the anti-HER2–targeted agent, which contributed to the increase in the number of long-term survivors and established the superiority of OS for the treatment of MRGC.
Electronic Supplementary Material
Supplementary materials are available at Cancer Research and Treatment website (https://www.e-crt.org).
Notes
Ethical Statement
This study protocol was approved by the Institutional Review Board of the Asan Medical Center, and informed consent was waived because of the retrospective nature of the study.
Author Contributions
Conceived and designed the analysis: Kang YK, Ryu MH, Koo DH.
Collected the data: Kang YK, Ryu MH, Chae H, Kim EJ, Moon MS.
Contributed data or analysis tools: Koo DH, Lee MY, Kang YK, Ryu MH, Chae H, Kim EJ, Moon MS.
Performed the analysis: Koo DH, Lee MY, Kang YK, Ryu MH.
Wrote the paper: Koo DH, Ryu MH, Kang YK.
Conflicts of Interest
Conflicts of interest relevant to this article was not reported.