Histologic Changes in Non–Small Cell Lung Cancer under Various Treatments: A Comparison of Histology and Mutation Status in Serial Samples
Article information
Abstract
Purpose
Histologic change is a resistant mechanism in lung cancer. The most common histological change is the switch from adenocarcinoma (AdenoCa) to small cell carcinoma (SCC) against to tyrosine kinase inhibitors (TKI). However, it is not clear whether other treatment modalities are involved in the histologic changes.
Materials and Methods
We investigated histological changes in eight cases after various treatments, and compared the molecular profiles between primary tumors and changed tumors using exome sequencing where tissue was available.
Results
Three cases of AdenoCa that were changed into SCC retained the initial mutations after TKI and/or surgical treatment. After treatment with TKI and immunotherapy, an EGFR (epidermal growth factor receptor)-mutant AdenoCa changed to squamous cell carcinoma (SqCa). SqCa in a patient treated with surgery was changed into combined AdenoCa and SqCa. These two cases showed the same genetic variations between the two distinct non–small cell carcinomas (NSCC). Three patients experienced two histologic changes, which the changed tumors returned to its original subtype or changed to a combined tumor after treatments. Four cases showed combined histology in the first or second change.
Conclusion
The histology of NSCC can be changed to a single pattern or combined subtypes after various treatment modalities, and the phenotypic changes seem not fixed. Therefore, additional morphologic changes may occur regardless of their genetic status and types of treatments. To refine the new treatment strategy, consecutive repeated biopsies in progressive disease or recurrent tumor are necessary.
Introduction
Despite dramatic response to tyrosine kinase inhibitors (TKI), drug resistance develops in many non–small cell carcinomas (NSCC) harboring epidermal growth factor receptor (EGFR) mutation within a median interval of a year [1,2]. Phenotypic change into small cell carcinoma (SCC) is a resistance mechanism in NSCC treated with TKI, and small cell transformation occurs 3% to 14% of patients with EGFR mutations [3,4]. Also, evolution into metastatic SCC under TKI treatment was reported [5]. This phenomenon is rare but has been reported more frequently in repeated biopsies after tumor recurrence. Previous studies reported the molecular determinants of lineage plasticity underlying this histologic change [6–9]. Transformed SCC after TKI treatment in EGFR-mutant adenocarcinoma (AdenoCa) branched out early from the clones with completely inactivated RB1 and TP53 [7]. RB1 and TP53 alterations represent early events in oncogenesis. EGFR-mutant lung cancers with RB1/TP53 alterations are at exceptionally high risk for SCC transformation [9]. Many pathologists postulate that the histologic change is due to the emergence of a subclone in a combined tumor. Such combined SCCs occur in approximately 1%–3% of biopsy specimens [10]. However, a high proportion of SCC (12%–26%) in surgical specimens shows NSCC components [10]. These data raise the question of whether or not TKI provokes the transformation to SCC. Recently, reports have shown that histologic change to squamous cell carcinoma (SqCa) is also a possible mechanism for resistance to TKI [11–13]. Histologic change was observed in anaplastic lymphoma kinase (ALK)-positive lung cancer without an EGFR mutation [14]. It is not clear whether other treatment modalities, in addition to TKI/ALK inhibitors, are involved in the histologic changes.
We conducted a retrospective study to investigate histological change in the resistant and recurrent tumors against various treatments. To determine if primary tumors and histologically changed tumors have a single origin or are two distinct tumors, we compared the molecular profiles between the primary and the histologically changed tumors using paired sample tissues.
Materials and Methods
1. Case selection
Between January 2010 and December 2019, we retrospectively collected the histologically changed lung cancers after treatment from the archives of pathology department at the Chungbuk National University Hospital. Eight consecutive cases were enrolled, and medical records were reviewed to extract clinicopathological data, including sex, age, smoking status, diagnoses, therapeutic agents, and survival. Two pathologists (Woo CG and Lee OJ) reviewed H&E and immunohistochemical slides. Tissues for six of the eight cases were available for additional molecular studies for next-generation sequencing (NGS). Also, we investigated survival after histologic change. Overall survival was the time from the initial diagnosis to death, and survival after histologic change was from the repeat biopsy to death.
2. Whole exome sequencing
We extracted DNA from sections of formalin-fixed, paraffin embedded tissues. For the detail sequencing methods, please see the Supplementary Methods.
Results
Table 1 and S1 Table show the clinicopathological characteristics of the eight patients. There were two females and six males with a median age of 71 years (range, 61 to 81 years). Five patients were heavy smokers. All patients undergoing histologic changes had NSCCs at initial diagnosis (six AdenoCas and two SqCas), and three patients showed two serial histologic changes after each treatment. Four patients had AdenoCa harboring EGFR mutations, which include three exon19 deletions and an exon20 missense mutation. Histologic switches were identified after receiving EGFR-TKI therapy. Three and two patients under-went surgery and immunotherapy, respectively, as initial treatments. Histologic change to SCC occurred in seven patients. The median time to change was 18 months (range, 7 to 40 months). Combined histologic features were observed in four tumors. The time to histologic change was shorter in patients with SqCa (7–10 months; median, 8.5 months) than in those with Adeno-Ca (12–40 months; median, 24 months).
1. SCC from AdenoCa
Histologic change to SCC took 14–31 months in five cases. Two cases were smokers, and three had advanced-stage cancer. Four patients (cases 1, 2, 4, and 7) with EGFR mutations were treated with EGFR-TKI, and the original EGFR mutation remained after histologic change in three tumors that were eligible for the molecular study (cases 1, 2, and 7). Two patients (cases 4 and 7) exhibited combined histology after histologic change. One patient (case 4) had combined SCC and AdenoCa at the same site, and one patient (case 7) exhibited combined SCC and SqCa in a metastasis at the lymph node (Fig. 1). In two early-stage cases (cases 1 and 3) resected by lobectomy, microscopic examination demonstrated pure AdenoCa at initial diagnosis, which underwent recurrence in the form of histologically changed SCC in distant sites (liver and brain). Both cases had no lymph node metastasis at initial diagnosis. An EGFR-wildtype tumor (case 3) was changed after complete resection as well. Whole exome sequencing analysis using available tissues (cases 1, 2, 3, and 7) found the same driver mutations in primary and histologically changed tumors (Fig. 2). The changed tumor retained identical mutations without newly acquired resistance mutations. Three cases (cases 1, 2, and 3) harbored TP53 and RB1 mutations in both primary and secondary tumors, irrespective of the EGFR mutation status. The EGFR-mutant lung cancer without TP53 and RB1 mutations (case 7) had APC, MED12, and RBL2 mutations in both the primary and changed tumors.

(A) Lung adenocarcinoma (AdenoCa; case 4) exhibiting a combined histology (small cell carcinoma [SCC] and AdenoCa) at histologic change under epidermal growth factor receptor tyrosine kinase inhibitor (EGFR-TKI) treatment. Squamous cell carcinoma (SqCa; case 5) in advanced stage exhibited histologic change into SCC after cisplatin plus radiotherapy. AdenoCa (case 7) harboring an EGFR exon19 deletion was changed into SqCC after EGFR-TKI plus programmed death-1 inhibitor treatment. Under another EGFR-TKI, the AdenoCa then histologically switched in the lymph node to a combined SCC and SqCC. After a surgical resection, the AdenoCa (case 8) histologically changed into SqCC in another lobe, and adenosquamous carcinoma was identified after a second resection. (B) Clinicopathological characteristics of the patients with histologically changed lung cancer. CCRT, concurrent chemoradiotherapy; CTx, chemotherapy; RTx, radiation therapy; TKI, tyrosine kinase inhibitor.
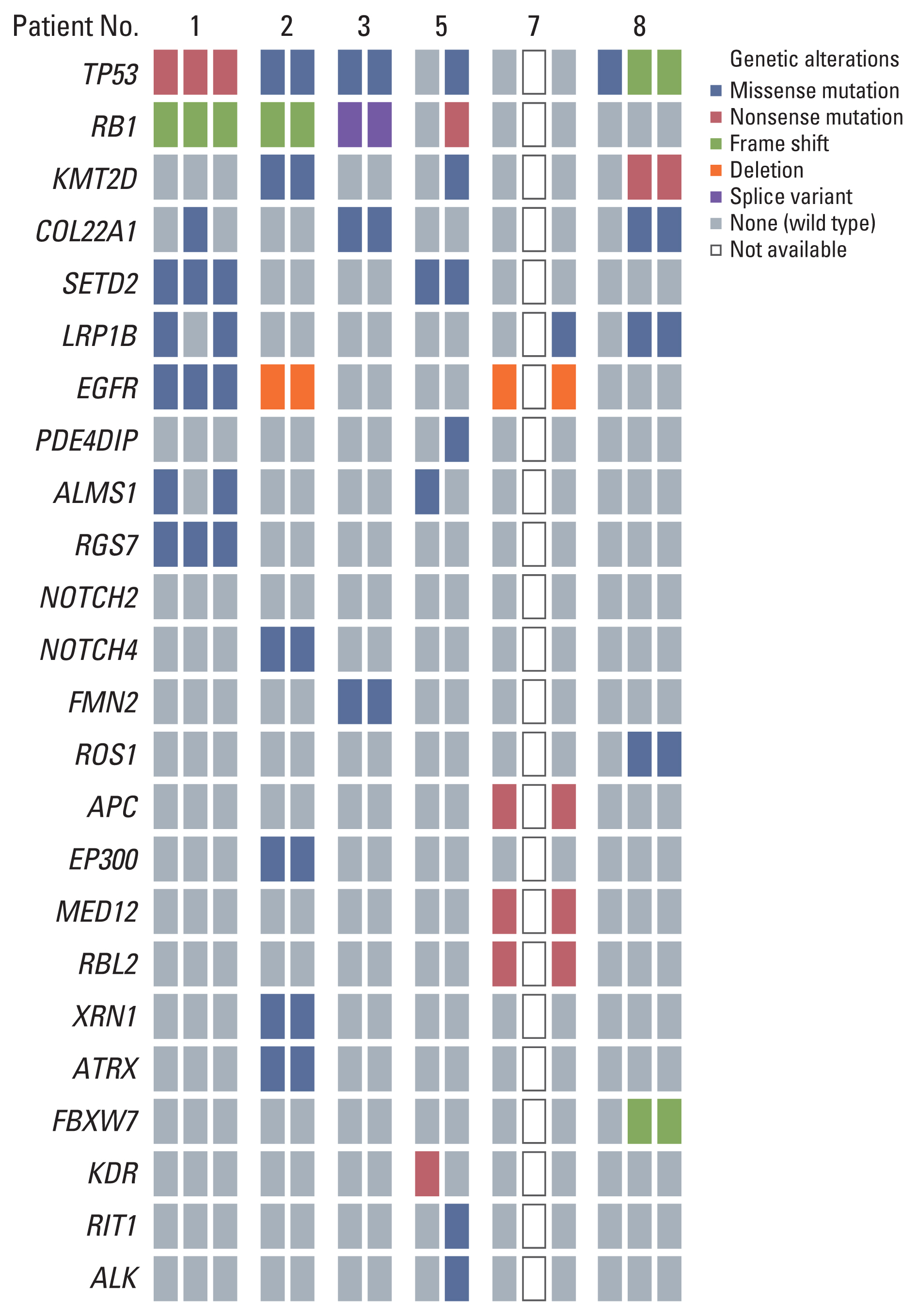
Whole exome sequencing analysis identified the same driver mutations between primary adenocarcinoma and changed small cell carcinoma (cases 1, 2, and 3). In case 5, the primary squamous cell carcinoma and changed small cell carcinoma had different mutations. Case 7 showed the same variants between primary adenocarcinoma and the third mixed small cell carcinoma and squamous cell carcinoma. In the molecular analysis of case 8, the molecular profile of the initial cell carcinoma and the third adenosquamous carcinoma. The same variants existed in the second and third tumors.
2. SCC from SqCa
Two patients (cases 5 and 6) with advanced-stage SqCa showed histologic change to SCC within 10 months after treatment (Fig. 1). Both were 61-year-old men and heavy smokers. Despite cisplatin and radiotherapy, one patient (case 5) suffered tumor progression to SCC as a new lesion in the contralateral lung. In the NGS study, there were different driver mutations between the first SqCa and the second SCC, and these tumors were independent of each other (Fig. 2). The other patient (case 6) with SqCa received immunotherapy (durvalumab and tremelimumab) plus chemotherapy (gemcitabine and carboplatin). At initial diagnosis, metastases in the liver and adrenal gland were clinically suspected based on radiology. Tumor progression in the liver was observed seven months after treatment, and the patient underwent a needle biopsy. A mixed tumor, including an SCC component, was observed. NGS was not carried out in this case, but clinically the tumors appeared to be the same.
3. SqCa from AdenoCa, and vice versa
Two patients (cases 7 and 8) demonstrated histologic changes (Fig. 1) between SqCa and AdenoCa after treatments. A non-smoking female (case 7) with an EGFR mutation received TKI therapy (gefitinib) and immunotherapy (nivolumab). Metastasis in the neck lymph node led to a repeated biopsy a year after treatment. Histological examination revealed SqCa, and the original EGFR mutation remained based on polymerase chain reaction (PCR) analysis. Another interesting case (case 8) showed two histologic changes after two complete resections. A smoking man with AdenoCa (pT3N0M0) underwent lobectomy. He has no recurrence for 40 months, but a new lesion was subsequently identified in the contralateral lung. The patient was diagnosed with SqCa (pT1aN0M0) after a secondary lobectomy and underwent four cycles of radiation. Unfortunately, the patient suffered from multiple lesions in the lung 14 months after the second operation. A needle biopsy specimen showed combined AdenoCa and SqCa. In the molecular analysis of this case, the molecular profile of the initial AdenoCa was different from both the second and the third tumors (Fig. 2). However, the second and third tumors kept the same mutations.
4. Two histologic changes
A patient with EGFR-mutant AdenoCa (case 1) was treated with complete resection. After seven months, recurrence in a lymph node was identified by radiology, and the patient received concurrent chemoradiotherapy with paclitaxel and cisplatin. The patient achieved a partial response but had another mass in the liver after eight months. He received TKI (afatinib), but the liver mass increased. This was the first histologic change to SCC in the liver to be identified. The patient subsequently experienced recurrence as a second change (AdenoCa) at the primary site (lung) after three cycles of etoposide and carboplatin to treat the changed SCC. The primary tumor and two changed tumors all had the same mutations (Fig. 2). Another patient with an EGFR-mutant AdenoCa (case 7) experienced change to SqCa in a lymph node twelve months after medical treatment (TKI and immunotherapy). This SqCa had the same EGFR mutation upon PCR analysis, but NGS analysis could not be performed because of the absence of available tissue. The patient received another TKI (YH25448) but suffered from tumor progression after four months. A third biopsy specimen showed combined SCC and SqCa in the lymph node. Both the primary tumor and the second changed combined tumor had the same mutations in the NGS study (Fig. 2).
Discussion
Histologic transformation to SCC was described as a resistant mechanism against TKI in EGFR-mutant lung AdenoCa [4]. This morphologic change to SCC occurs in a subset of patients with concurrent inactivation of TP53 and RB1. It is associated with large-scale genomic alterations, including an APOBEC mutation signature [6,7]. Patients with AdenoCa harboring EGFR/RB1/TP53 mutations have the potential to be in SCC transformation after TKI treatment [9]. In our study, two patients (cases 1 and 2) went through this course. Case 1 showed metastatic SCC in the liver after TKI plus complete surgical resection. The transformed SCCs kept the initial EGFR mutation, consistent with previous reports [4,11,12]. Both primary AdenoCa and secondary SCC components shared the same mutations in the NGS study, indicating that the SCC directly originated from the AdenoCa. Both the transformed SCC and the initial AdenoCa had inactivated RB and TP53, suggesting that complete inactivation of these genes predicts transformation to SCC, as reported in a previous study [7]. However, we also observed that histologic changes to SCC are not always associated with EGFR/RB1/TP53 mutations. Histologic change to SCC was demonstrated after complete resection for the tumor without an EGFR mutation (case 3). EGFR-mutant AdenCa without RB1 and TP53 mutations (case 7) was also changed to SCC as combined histology (SqCa). This patient had APC, MED12, and RBL2 mutations in both the primary and changed tumors. Not only RB1/TP53 mutations but also APC/MED12/RBL2 mutations can contribute to the histologic change to SCC. Furthermore, it is considered that this phenotypic change from AdenoCa to SCC is not fixed or one-way (case 1). The second recurrent tumor of case 1 morphologically returned to its original histology (AdenoCa) after chemotherapy for SCC.
NSCC that are histologically changed after treatment may not be destined to become SCC. NSCC can change to another type of NSCC, from AdenoCa to SqCa, and vice versa [15,16]. After TKI and PD-1 inhibitor treatment, one of the EGFR-mutant AdenoCa was changed into SqCa, which had the same EGFR mutation (Exon19 deletion) as the primary AdenoCa (case 7). The AdenoCa in the last patient (case 8) underwent two complete histologic changes between AdenoCa and SqCa after two complete resections (AdenoCa → SqCa → combined AdenoCa and SqCa). In the NGS study of these three tumors, except for the first tumor, the other two lesions had the same mutation profiles. Only the TP53 mutation was identified in the first tumor, which has the potential for false negatives for other genes because the tumor sample (a 5-year-old formalin-fixed paraffin-embedded block) was old. However, the TP53 mutation (missense) in the first tumor was different (frameshift) from the other two. These results indicate that the second and third tumors were of the same origin, and SqCa can be changed into AdenoCa after surgery and radiation therapy. SqCa changed from AdenoCa can be an acquired resistance after EGFR-TKI therapy [15]. However, to our knowledge, there are no cases of AdenoCa changed from SqCa after surgery in lung cancer.
One potential explanation for histologic change is the co-existence of diverse tumor cells in the original tumor. The possibility of a mixed tumor in the initial diagnosis cannot be excluded because tumor volumes from needle biopsies are limited. Tumor heterogeneity could contribute to histologic switches allowing carcinoma components to survive against treatments. The first and second tumors of case 7 cannot rule out the possibility of adenosquamous carcinoma because of small biopsies with limited information. On the other hand, combined tumors in this study may represent the plasticity of cancer stem cells showing histologic change of cancer cells (from AdenoCa to SqCa, and vice versa). The third tumor of case 8 is likely to be changed to adenosquamous carcinoma or combined AdenoCa and SqCa from the second SqCa. In other words, the identity of these second and third tumors is another primary tumor of the same origin, different from histology and molecular profiles of the first tumor.
Our data showed that surgery could also act as a selective pressure. The spatial pressure such as surgery, and the affected organ in recurrent or progressive tumors might be influenced by the local microenvironment. If the tumor cells survive after complete resection, they live only in the circulation and they may be attacked by the immune system of blood stream (cases 1, 3, and 8). These pressures may also cause histologic changes and tumor cells settle in other organs in a changed histology. Histologic change from AdenoCa to SCC after surgery and adjuvant therapy has been previously described [17]. One patient (case 3) in our study is interesting because the diagnosis of primary lung cancer and its brain metastasis was confirmed by surgical resection, and both were pure AdenoCa and SCC, respectively. This is the first report about histologic change to SCC after only surgery for lung AdenoCa, regardless of the EGFR mutation.
In our study, two patients with SqCa also showed phenotypic switches to SCC. The NGS study in case 5 showed that the changed SCC lost the existing mutations and presented new mutations. This histologic change reflects two different tumors. Another patient (case 6) had metastatic combined SCC having SqCa component in the liver after immunotherapy and chemotherapy for lung SqCa. Because the NGS study in this patient was not available, the exact relationship between the two tumors was unclear.
We investigated the clinicopathological features of NSCCs showing histologic changes and compared the molecular profiles between the primary lung tumor and the changed tumor after various treatments. Our study suggests that NSCC subjected to various treatments can undergo histologic changes to SCC and/or another type of NSCC, independent of treatment modalities and molecular characteristics (Fig. 3). NSCC widens the spectrum of histologic evolution as a resistance mechanism. Genetic diversity within the same tumor [18] led to histologic change through extrinsic (various treatments) and intrinsic factors (competition among tumor cells or between tumor cells and immune cells). Furthermore, the tumors that once experienced histologic change can change again. Whenever progressive disease or a recurrent tumor is identified, repeated biopsies are necessary to refine the treatment strategy. This study was conducted on a small number of cases, and the genetic phenotypes were confirmed in matched tumors. Therefore, further research, including a larger number of patients, is required.
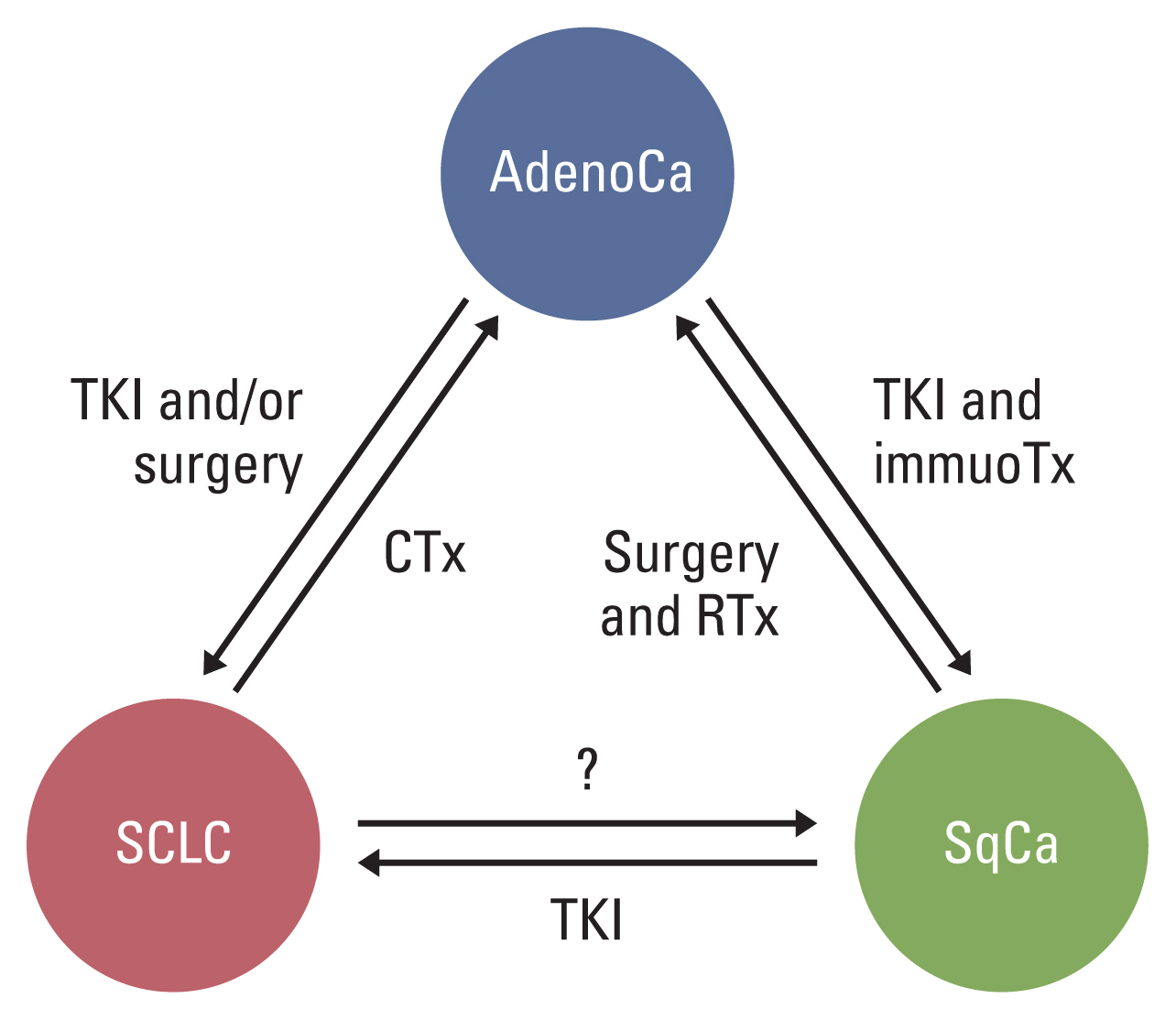
Non–small cell carcinoma subjected to various selective pressures can undergo histologic change to small cell carcinoma or another non-small cell carcinoma, independent of treatment modalities. AdenoCa, adenocarcinoma; CCRT, concurrent chemoradiotherapy; CTx, chemotherapy; immunoTx, immunotherapy; SCLC, small cell lung cancer; SqCa, squamous cell carcinoma; TKI, tyrosine kinase inhibitor.
Electronic Supplementary Material
Supplementary materials are available at Cancer Research and Treatment website (https://www.e-crt.org).
Notes
Ethical Statement
The study was approved by the Institutional Review Board of Chungbuk National University Hospital (Cheongju, Republic of Korea, IRB No. 2018-08-001). The patient provided informed consent in accordance with the Declaration of Helsinki.
Author Contributions
Conceived and designed the analysis: Woo CG, Son SM, Lee HC, Han HS, Kim D, Lee OJ.
Collected the data: Woo CG, Son SM, Lee HC, Han HS, Lee KH, Kim D, Lee OJ.
Contributed data or analysis tools: Woo CG, Son SM, Lee KH, Kim EG. Lee OJ.
Performed the analysis: Woo CG, Son SM, Lee KH, Kim EG. Lee OJ.
Wrote the paper: Woo CG, Son SM, Lee OJ.
Molecular analysis: Woo CG, Son SM, Kim EG.
Conflicts of Interest
Conflict of interest relevant to this article was not reported.
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MISP) (2020R1A5A2017476).

