Real-World Efficacy Data and Predictive Clinical Parameters for Treatment Outcomes in Advanced Esophageal Squamous Cell Carcinoma Treated with Immune Checkpoint Inhibitors
Article information
Abstract
Purpose
This study aimed to evaluate the real-world efficacy of immune checkpoint inhibitors (ICIs), and to identify clinicolaboratory factors to predict treatment outcomes in patients with advanced esophageal squamous cell carcinoma (ESCC) receiving ICIs.
Materials and Methods
Sixty patients with metastatic or unresectable ESCC treated with nivolumab (n=48) or pembrolizumab (n=12) as ≥ second-line treatment between 2016 and 2019 at Asan Medical Center were included.
Results
The median age of the patients was 68 years (range, 52 to 76 years), and 93.3% were male. Most patients had metastatic disease (81.7%) and had been previously treated with fluoropyrimidines, platinum, and taxane. In 53 patients with measurable disease, the overall response rate and disease control rate were 15.1% and 35.8%, respectively. With a median follow-up duration of 16.0 months, the median progression-free survival (PFS) and overall survival (OS) were 1.9 months (95% confidence interval [CI], 1.54 to 2.19) and 6.4 months (95% CI, 4.77 to 8.11), respectively. After multivariate analysis, recent use of antibiotics, low prognostic nutrition index (< 35.93), high Glasgow Prognosis Score (≥ 1) at baseline, and ≥ 1.4-fold increase in neutrophil-to-lymphocyte ratio after one cycle from baseline were significantly unfavorable factors for both PFS and OS. Younger age (< 65 years) was a significant factor for unfavorable PFS and hyponatremia (< 135 mmol/L) for unfavorable OS.
Conclusion
The use of ICIs after the failure of chemotherapy showed comparable efficacy in patients with advanced ESCC in real practice; this may be associated with host immune-nutritional status, which could be predicted by clinical and routine laboratory factors.
Introduction
Esophageal carcinoma is one of the most common cancers worldwide and its prevalence is highest in East Asia [1]. Esophageal squamous cell carcinoma (ESCC) is the predominant histological subtype in East Asia and accounts for more than 90% of all esophageal carcinomas in Korea [2,3]. Despite the gradual improvement in survival, the 5-year relative survival rate remains only 37.4% according to the nationwide cancer statistics in Korea; this is because most of the patients usually have an advanced stage at the time of diagnosis [2]. For unresectable or metastatic esophageal cancer, palliative chemotherapy is recommended and the most commonly used first-line regimens are based on a combination of fluoropyrimidine and platinum [4], while taxane and irinotecan have also shown antitumor activity as salvage therapies [5,6]. Nevertheless, the median survival for patients with unresectable or metastatic esophageal carcinoma remains 8–10 months, and the development of novel therapeutic agents are urgently required.
Following several phase I/II studies showing promising results of immune checkpoint inhibitors (ICIs) against programmed cell death 1 (PD-1), including pembrolizumab and nivolumab [7–9], the phase III KEYNOTE-181 [10] and ATTRACTION-3 [11] trials have demonstrated better efficacy and safety with ICIs in patients with esophageal carcinoma after failure of first-line therapy compared with conventional chemotherapy. In the KEYNOTE-181 trial, two thirds of patients had ESCC subtype, and pembrolizumab significantly improved the overall survival (OS) in patients with programmed cell death ligand 1 (PD-L1) positive tumors (defined as a combined positive score [CPS] ≥ 10) compared with conventional chemotherapy with docetaxel, paclitaxel, or irinotecan. The ATTRACTION-3 trial enrolled only patients with ESCC and nivolumab was demonstrated to significantly improve OS, regardless of PD-L1 status, compared with conventional chemotherapy of docetaxel or paclitaxel. Based on these results, pembrolizumab and nivolumab were approved by the U.S. Food and Drug Administration for ≥ second-line treatment in patients with advanced ESCC. Besides survival benefits, ICIs demonstrated better safety than cytotoxic chemotherapy (treatment-related grade ≥ 3 adverse events: 18.2% for pembrolizumab vs. 40.9% for chemotherapy and 18% for nivolumab vs. 63% for chemotherapy); this is particularly important for patients with esophageal cancer seen in routine clinical practice because they are commonly elderly with malnutrition and multiple comorbidities [12].
However, the clinical benefit of ICIs may be limited to only a small portion of patients with advanced esophageal carcinoma, and a subset of patients might experience more rapid tumor progression during the first several months of ICIs compared with chemotherapy as observed in the ATTRACTION-3 study. This suggests the importance of identifying biomarkers to predict which patients could benefit from ICIs. Therefore, this study aimed to evaluate the real-world efficacy of ICIs and to identify clinicolaboratory factors to predict treatment outcomes in patients with advanced ESCC receiving ICIs.
Materials and Methods
1. Patients
Using the electronic medical records database system, a total of 99 patients were identified based on the diagnosis of ESCC and administration history of ICIs between 2015 and 2019 at Asan Medical Center, Seoul, Republic of Korea. Among them, nine patients were given ICIs for other malignancies along with the past history of early ESCC, 10 patients were treated with ICIs as neoadjuvant or adjuvant therapy, and 20 patients were treated with blinded drugs (ICIs vs. placebo) in clinical trials for ESCC. Excluding those patients, 60 patients with metastatic or unresectable ESCC treated with nivolumab (n=48) or pembrolizumab (n=12) as ≥ second-line therapy between May 2016 to December 2019 were included in this study. Patients received nivolumab 3 mg/kg intravenously every 2 weeks or pembrolizumab 2 mg/kg intravenously every 3 weeks until disease progression, unacceptable toxicity, or patients’ refusal. The tumor response was assessed using computed tomography every 6 to 8 weeks based on Response Evaluation Criteria in Solid Tumors (RECIST) criteria ver. 1.1 [13].
2. Data collection
Clinical and laboratory data were collected, and baseline characteristics included the following: patient demographics (age, sex, smoking and alcohol history, and Eastern Cooperative Oncology Group [ECOG] performance status), disease status (initially metastatic, recurrent metastatic, recurrent localized unresectable, or localized unresectable), sites of metastasis (lymph node, liver, lung, bone, peritoneum, and others), the recent use of antibiotics, which was defined as at least one dose of antibiotics within a month before ICI administration, and laboratory findings. Peripheral blood tests were performed at baseline before initiation of ICIs and before each treatment cycle, from which the absolute neutrophil count, absolute lymphocyte count, hemoglobin, platelet, sodium, albumin, lactate dehydrogenase (LDH), and C-reactive protein (CRP) values were collected. The neutrophil-to-lymphocyte ratio (NLR) was calculated by dividing the absolute neutrophil count by the absolute lymphocyte count, and the platelet-to-lymphocyte ratio (PLR) was calculated by dividing the absolute platelet count by the absolute lymphocyte count [14]. The prognostic nutrition index (PNI) was calculated using the following formula: 10×serum albumin (g/dL)+0.005×total lymphocyte count (/mm3) [15]. The Glasgow Prognosis Score (GPS) was calculated as follows: patients with increased CRP (> 10 mg/L) and hypoalbuminemia (< 3.5 g/dL) were allocated a score of 2, patients with only one of these laboratory abnormalities were allocated a score of 1, and patients with neither were allocated a score of 0 [16].
3. Available tissue collection
Available archival tissues from primary or metastatic sites were used to evaluate the PD-L1 status. Immunohistochemistry (IHC) staining was performed on a Dako Autostainer Link 48 system (Agilent Technologies, Santa Clara, CA) using a Dako PD-L1 IHC 22C3 pharmDx kit (Agilent Technologies). The level of PD-L1 protein expression was determined using the TPS or CPS, which was calculated as the number of PD-L1-stained cells (TPS; tumor cells or CPS; tumor cells, lymphocytes, and macrophages) divided by the total number of viable tumor cells and multiplied by 100. Tumor PD-L1 positivity was defined as TPS ≥ 1% or CPS ≥ 1 and also analyzed according to the various cutoff values (1, 5, and 10).
4. Statistical analysis
Categorical and quantitative data were compared using the chi-square test or Fisher exact test and Mann-Whitney U-test, respectively. The percentage change in laboratory values from baseline was calculated using the following formula: 100×([post-cycle 1 values–baseline value]/baseline value). The cutoff values for low vs. high baseline NLR, PLR, or PNI or the percentage changes of NLR and PLR were determined by receiver operating characteristic curves for early disease progression at the first tumor evaluation. Reference ranges were used as the cutoff values for other laboratory findings (hemoglobin, sodium, albumin, LDH, and CRP). Progression-free survival (PFS) was calculated from the date of initiation of ICIs to the date of disease progression or death from any cause, whichever occurred first. OS was calculated from the date of initiation of ICIs to the date of death from any cause. Survival was estimated using the Kaplan-Meier method, and the log-rank test was used to compare differences between the curves. Prognostic factors for PFS and OS were analyzed using Cox proportional hazard regression model. A logistic regression model was used to calculate the odds ratios (ORs) of the objective response rate (ORR) (which denoted the proportion of patients with a complete response [CR] or partial response [PR]) and the disease control rate (DCR) (the proportion of patients with a CR, PR, or stable disease [SD]) according to clinical and laboratory factors. Multivariate analysis included factors that were significant (p < 0.1) in the univariate analysis, and the backward elimination model was used in the multivariate analysis. The NLR was used instead of absolute neutrophil or lymphocyte counts, and the GPS and PNI were used instead of CRP and albumin to avoid multicollinearity in multivariate analysis of the Cox proportional hazard regression and logistic regression models. A two-sided p-value < 0.05 was considered significant, and all statistical analyses were performed using the SPSS ver. 25.0 software package (IBM Corp., Armonk, NY).
Results
1. Patient characteristics
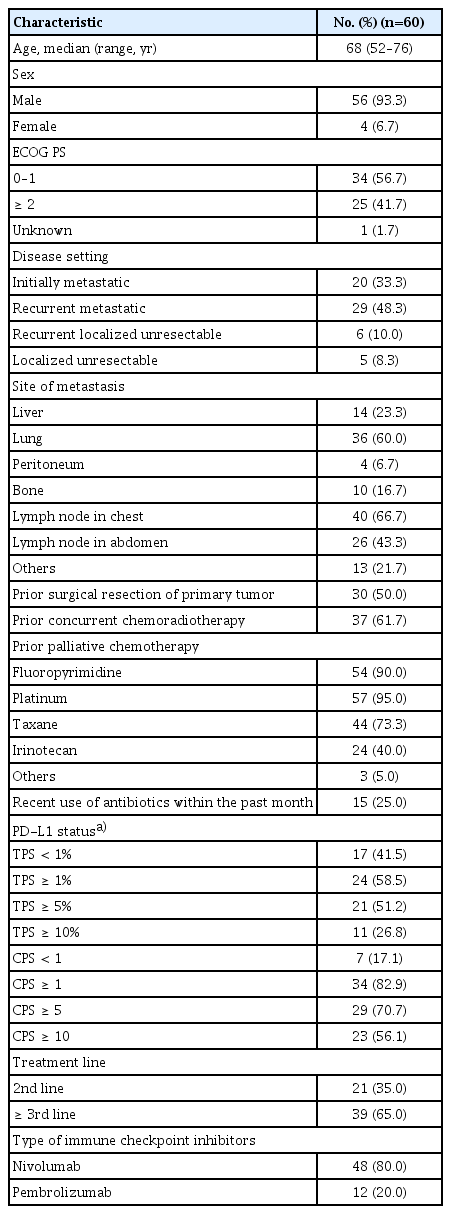
Table 1 summarizes the baseline characteristics of the study patients. Sixty patients received nivolumab (n=48, 80.0%) or pembrolizumab (n=12, 20.0%) as second-line (35.0%) or ≥ third-line (65.0%) therapy. The median age of the patients was 68 years (range, 52 to 76 years), and 93.3% of the patients were male. Most patients had metastatic disease (81.7%) and been previously treated with fluoropyrimidines, platinum, and taxane. Fifteen patients (25.0%) received at least one dose of antibiotics in the month before ICI administration due to respiratory tract infection (n=7), gastrointestinal and biliary tract infection (n=6), and as a prophylactic (n=2). There were no significant differences in ECOG performance status (≥ 2) (36.8% vs. 45.0%) or hypoalbuminemia (42.1% vs. 39.0%) between patients with and without recent use of antibiotics. Among 60 patients, PD-L1 status was evaluated in 41 patients (68.3%) with available tissues receiving nivolumab (n=32, 53.3%) or pembrolizumab (n=9, 15.0%). The PD-L1 positivity was 58.5% by TPS ≥ 1% and 82.9% by CPS ≥ 1.
2. Efficacy outcomes
S1 Table summarizes the objective tumor response to ICIs. In 53 patients with measurable disease, CR, PR, and SD were achieved in two (3.8%), six (11.3%), and 11 (20.8%) patients, respectively, and 30 patients (56.6%) showed progressive disease (PD); the ORR and DCR were 15.1% (95% confidence interval [CI], 7.6 to 27.3) and 35.8% (95% CI, 24.3 to 49.3). Representative images of two CR responders are shown in Fig. 1. In seven patients with non-measurable disease, four (57.1%) had non-CR/non-PD and two (28.6%) had PD; DCR was 57.1% (95% CI, 25.0 to 84.3). At a median follow-up duration of 16.0 months (95% CI, 7.4 to 24.5), the median PFS and OS were 1.9 months (95% CI, 1.54 to 2.19) and 6.4 months (95% CI, 4.77 to 8.11), respectively (Fig. 2).
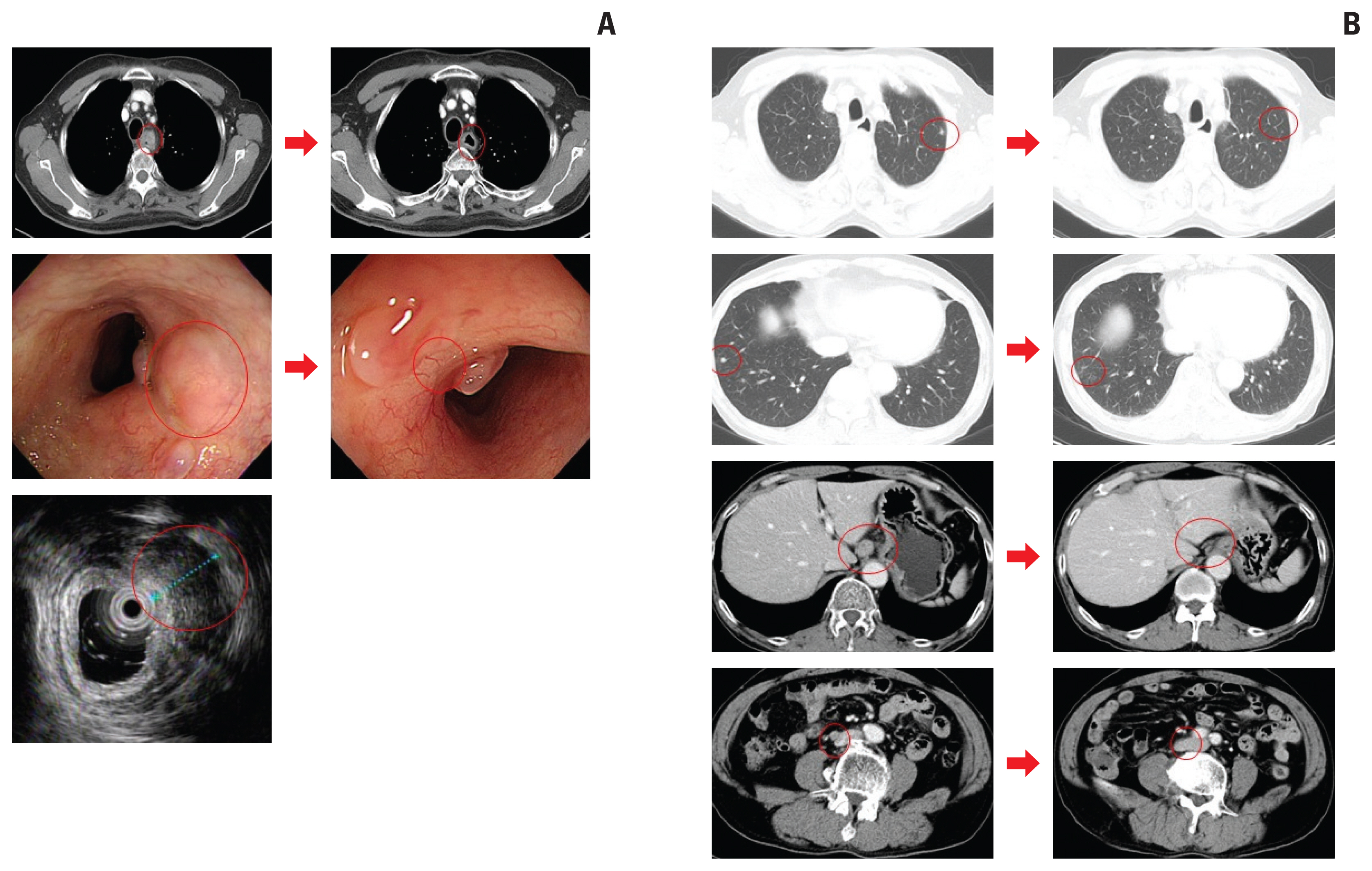
Representative images of two patients who achieved complete response (CR) with immune check point inhibitors. (A) Esophagogastroduodenoscopy and endoscopic ultrasound images of a 71-year-old man with recurrent localized unresectable esophageal squamous cell carcinoma after chemoradiotherapy who was treated with pembrolizumab. After completion of 2-year pembrolizumab treatment, endoscopically CR with no residual cancer cells on biopsy was confirmed. (B) Computed tomography scans of 61-year-old man with recurrent metastatic esophageal squamous cell carcinoma who was treated with nivolumab. After 6 cycles of nivolumab treatment, metastatic lymph nodes and lung nodules disappeared.

The Kaplan-Meier curves of progression-free survival (A) and overall survival (B). CI, confidence interval.
There were no significant differences in ORR and DCR between nivolumab and pembrolizumab treatment in 53 patients with measurable disease (ORR, 15.9% vs. 11.1% 11.1%; p > 0.99 and DCR, 31.8% vs. 55.6%; p=0.282). There were no significant differences in PFS (p=0.138) and OS (p=0.633) between nivolumab and pembrolizumab treatment.
3. Analysis of potential clinicolaboratory factors for PFS and OS
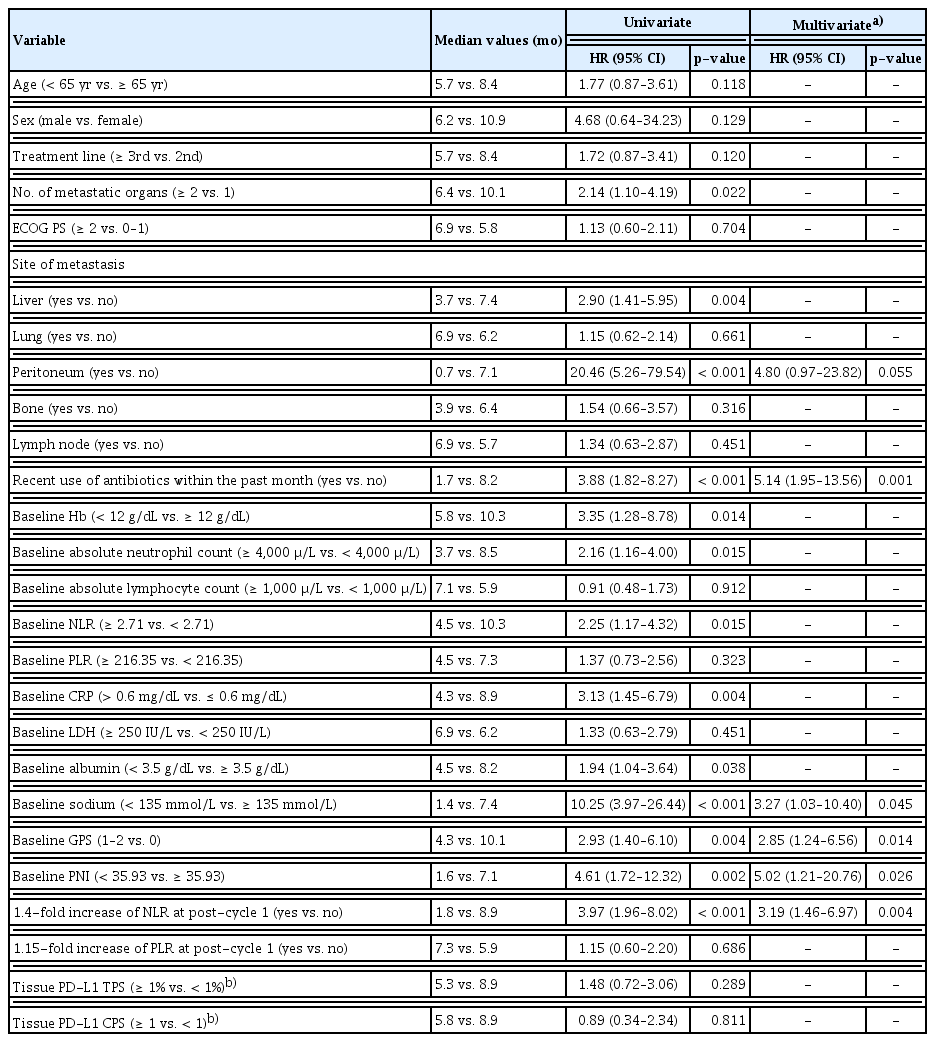
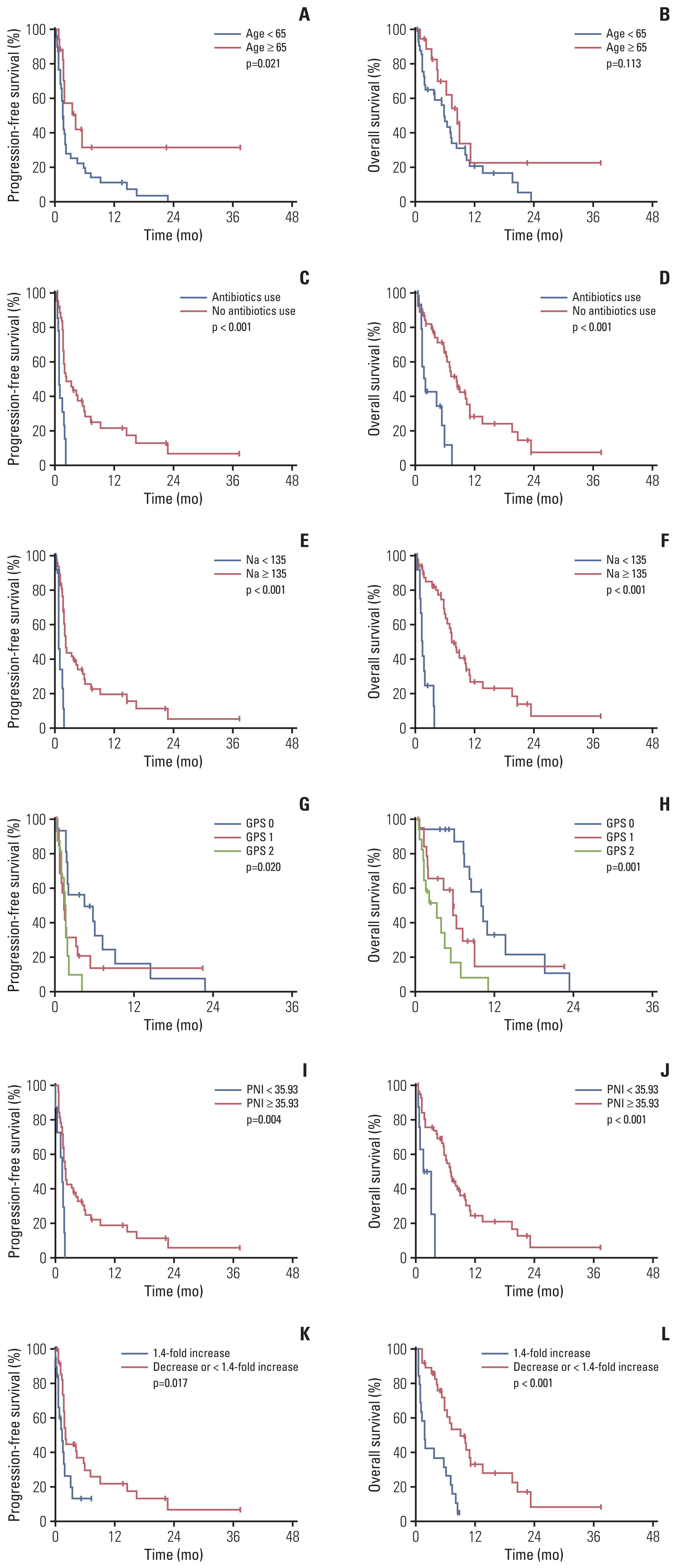
Table 2 summarizes the univariate and multivariate analyses for PFS. After multivariate analyses, younger age (< 65 years) (hazard ratio [HR], 3.94; 95% CI, 1.58 to 9.85; p=0.003), recent use of antibiotics within the past month (HR, 4.32; 95% CI, 1.81 to 10.32; p=0.001), baseline GPS (≥ 1) (HR, 2.43; 95% CI, 1.81 to 10.32; p=0.041), baseline low PNI (< 35.93) (HR, 4.07; 95% CI, 1.29 to 12.90; p=0.017) and a 1.4-fold increase in NLR from baseline to post-cycle 1 (HR, 2.68; 95% CI, 1.18 to 6.09; p=0.019) were significantly associated with poor PFS. Table 3 summarizes univariate and multivariate analyses for OS. After multivariate analyses, recent use of antibiotics in the past month (HR, 5.14; 95% CI, 1.95 to 13.56; p=0.001), hyponatremia (< 135 mmol/L) (HR, 3.27; 95% CI, 1.03 to 10.40; p=0.045), baseline GPS (≥ 1) (HR, 2.85; 95% CI, 1.24 to 6.56; p=0.014), baseline low PNI (< 35.93) (HR, 5.02; 95% CI, 1.21 to 20.76; p=0.026), and a 1.4-fold increase in NLR from baseline to post-cycle 1 (HR, 3.19; 95% CI, 1.46 to 6.97; p=0.004) were significantly associated with poor OS. The Kaplan-Meier curves of PFS and OS according to significant factors in the multivariate analysis are shown in Fig. 2. PFS and OS curves according to clinicolaboratory factors that were significantly associated with PFS or OS only in the univariate analysis are shown in S2 Fig.
4. Analysis of potential clinicolaboratory factors for objective tumor response
The univariate and multivariate analyses for ORR according to potential clinicolaboratory factors are summarized in S3 Table. After multivariate logistic regression, baseline GPS (≥ 1) was marginally associated with lower ORR (OR, 0.19; p=0.078) and significantly associated with lower DCR (OR, 0.20; p=0.027). In univariate analysis for ORR and DCR, lower ORR and DCR were associated with the recent use of antibiotics within the past month (ORR, 0% vs. 19.0%; OR, not calculated and DCR, 15.4% vs. 50.0%; OR, 0.17; p=0.040), high baseline NLR (≥ 2.71) (ORR, 6.1% vs. 27.3%; OR, 0.17; p=0.046 and DCR, 30.3% vs. 59.1%; OR, 0.30; p=0.037), increased baseline CRP (> 0.6 mg/dL) (ORR, 2.9% vs. 35.7%; OR, 0.05; p=0.008 and DCR, 34.3% vs. 57.1%; OR, 0.39; p= 0.147), and baseline GPS (≥ 1) (ORR, 6.1% vs. 25%; OR, 0.19; p=0.078 and DCR, 30.3% vs. 62.5%; OR, 0.26; p=0.036).
5. Analysis of changes in laboratory factors between baseline and post-cycle 1 for PFS and OS
Among 60 patients, the hemoglobin, albumin, and Na values, as well as the NLR and PLR before the second cycle were available in 55 patients, and the CRP and LDH values before the second cycle were available in 44 patients. Patients with a 1.4-fold increased NLR at post-cycle 1 had significantly worse PFS (p=0.017) and OS (p < 0.001) than those with a decreased or < 1.4-fold NLR at post-cycle 1 (Fig. 3K and L). S4 Fig. shows PFS and OS according to changes in laboratory factors. Patients with hypoalbuminemia at post-cycle 1 had significantly worse PFS (p=0.038) and OS (p < 0.001) than those with normal albumin at post-cycle 1. Patients with hyponatremia at baseline or post-cycle 1 had significantly worse PFS (p < 0.001) and OS (p < 0.001) than those without hyponatremia at both. Patients without increased CRP at both had significantly better PFS (p=0.023) and OS (p=0.010) than other patients. There were no significant differences in PFS and OS according to changes in hemoglobin, LDH, and PLR.
6. Analysis of tissue PD-L1 status for the efficacy outcomes
The ORR and DCR were not associated with tissue PD-L1 positivity using TPS or CPS (S3 Table). There were also no significant differences in PFS and OS according to PD-L1 positivity (S5 Fig.). According to PD-L1 TPS, median PFS and OS were 1.6 months and 5.3 months in PD-L1 TPS ≥ 1%, whereas 1.7 months and 8.9 months in PD-L1 TPS < 1% (p=0.532 and p=0.285, respectively). According to PD-L1 CPS, median PFS and OS were 1.7 months and 5.8 months in PD-L1 CPS ≥ 1, whereas 1.6 months and 8.9 months in PD-L1 CPS < 1 (p=0.128 and p=0.811, respectively). Likewise, no significant differences in PFS and OS were observed according to PD-L1 positivity (TPS ≥ 5%, 10% or CPS ≥ 5, 10).
Discussion
Although the treatment options in patients with advanced esophageal carcinoma have expanded through the use of ICIs, the efficacy of ICIs in real practice and the value of biomarkers to identify optimal candidates remains unclear. The current study confirmed the efficacy of ICIs in patients with advanced ESCC after failure of prior chemotherapy in real practice. Moreover, we comprehensively evaluated the prognostic values of potential clinicolaboratory factors for the efficacy of ICIs. Our results showed comparable ORR (15.1%), median PFS (1.9 months), and median OS (6.4 months) to those of the previous KEYNOTE-181 [10] and ATTRACTION-3 trials [11]. After multivariate analysis, recent use of antibiotics, low PNI (< 35.93) and high GPS (≥ 1) at baseline, and ≥ 1.4-fold increase in NLR from baseline to post-cycle 1 were significantly unfavorable factors for both PFS and OS. Younger age (< 65 years) was a significant factor for unfavorable PFS and hyponatremia (< 135 mmol/L) for unfavorable OS.
The KEYNOTE-181 trial [10] compared second-line pembrolizumab with conventional chemotherapy in patients with unresectable or locally advanced esophageal carcinoma (ESCC in two thirds, adenocarcinoma in one third). Among three analysis populations (all patients, patients with ESCC, patients with PD-L1 CPS ≥ 10), only patients with PD-L1 CPS ≥ 10 showed significant OS benefits with pembrolizumab compared to chemotherapy (HR, 0.69; p=0.007), whereas patients with ESCC showed a non-significant trend toward better OS with pembrolizumab. Overall PFS was also better with pembrolizumab than chemotherapy (HR, 0.73; 95% CI, 0.54 to 0.97). The difference in ORR between patients who received pembrolizumab and those who received chemotherapy was greater in the PD-L1+ population (21.5% vs. 6.1%, p < 0.001) than in the ESCC population (16.7% vs. 7.4%, p=0.002) or in the total patient population (13.1% vs. 6.7%, p=0.004). In contrast, in the ATTRACTION-3 trial [11], in which only patients with ESCC were enrolled, second-line nivolumab treatment showed superior OS compared to chemotherapy and survival benefit was not significantly affected by PD-L1 expression status at various cutoffs. Nevertheless, the HR of nivolumab for OS, compared with chemotherapy, was 0.69 (95% CI, 0.51 to 0.94) in patients with PD-L1 ≥ 1%, whereas 0.84 (95% CI, 0.62 to 1.14) in patients with PD-L1 < 1%. Although pembrolizumab and nivolumab demonstrated survival benefits in advanced esophageal carcinoma, not all patients could benefit from them. Rather, a subset of patients experienced more rapid disease progression during an early period of nivolumab treatment compared to those who received chemotherapy; a shorter median PFS with nivolumab than with chemotherapy (1.7 months vs. 3.4 months) in the ATTRACTION-3 trial [11]. Therefore, the identification of biomarkers for the efficacy and/or resistance to ICIs is essential to determine whether an individual patient should be treated with ICIs or chemotherapy as a preferred salvage therapy. Although PD-L1 was suggested as a biomarker for pembrolizumab in the KEYNOTE-181 trial, patients with low or undetectable PD-L1 expression may still gain clinical benefit from pembrolizumab, while a considerable proportion of patients with high PD-L1 expression may not [10]. Furthermore, the PD-L1 positivity was not associated with the efficacy outcomes in our study, even though it was limited by the small sample size. Therefore, the current study focused on clinical and routine laboratory parameters other than PD-L1 to identify predictive biomarkers for ICIs in ESCC.
Recently, several studies have highlighted the utility of immune prognostic scores to shed light on the importance of routine laboratory parameters because they are easily accessible and sensitive inflammatory- or nutrition-based biomarkers [17,18]. Although these biomarkers, including CRP, GPS, PNI, and NLR, have shown prognostic value in various treatment settings for diverse cancers including esophageal cancer [14,18–22], they have gained more attention recently with regard to immune-oncology treatment. Our study demonstrated that baseline high GPS and low PNI, which means high CRP and low lymphocyte counts, respectively, and commonly low albumin levels, could be potential predictors for worse PFS and OS in patients with ESSC treated with PD-1 inhibitors. CRP has been known to be elevated by pro-inflammatory cytokines such as interleukin (IL)-1, IL-8, and IL-6, and has a profound suppressive effect on adaptive immunity by impacting both effector T cells and antigen presentation. Moreover, CRP is associated with a poor clinical outcome for various cancers treated with ICIs, including melanoma and non–small cell lung cancer (NSCLC) [21]. Hypoalbuminemia has also been associated with the impairment of systemic cell-mediated immune responses, such as macrophage activation and granuloma formation, as well as with poor prognosis in patients with cancer receiving various treatments [17,18,20]. Furthermore, a baseline peripheral blood absolute lymphocyte count ≥ 1,000/μL has been shown to be significantly associated with favorable PFS (HR, 0.55; p=0.04) and OS (HR, 0.36; p=0.03) in patients with NSCLC treated with nivolumab [23]. Besides each blood-based marker, their combination indexes such as GPS, PNI, and NLR have also shown predictive value for ICIs. In our study, the GPS and PNI had better performance in predicting treatment outcomes than CRP, albumin, and absolute lymphocyte counts. Kurosaki et al. [24] reported that a higher GPS was significantly associated with a shorter PFS (median, 3.0 months vs. 1.6 months vs. 1.4 months for a GPS of 0 vs. 1 vs. 2, respectively; p=0.005) and OS (median, 11.0 months vs. 5.1 months vs. 2.9 months for a GPS of 0 vs. 1 vs. 2, respectively; p < 0.001) in 80 patients with advanced gastric cancer treated with nivolumab. Kurosaki et al. [24] also reported that a high baseline NLR, indicating neutrophilia and lymphopenia, and low PNI were significantly associated with worse PFS and OS in 102 patients with advanced NSCLC treated with anti-PD-1 inhibitors (median PFS, 3.2 months for the high-NLR group vs. 7.3 months for the low-NLR group, p=0.009, and 3.3 months for the low-PNI group vs. 6.3 months for the high-PNI group, p=0.007; median OS, 3.7 months for the high-NLR group vs. 9.8 months for the low-NLR group, p=0.002, and 4.2 months for the low-PNI group vs. 11.5 months for the high-PNI group, p < 0.001) [25]. Interestingly, in our study, the NLR played a role as a predictor of PFS and OS when considered as a change after one cycle of ICIs, but not the baseline value (which was significant in the univariate analysis for PFS and OS, but not in the multivariate analysis). A 1.4-fold increase in the NLR from baseline was significantly associated with poorer PFS and OS. The consensus defining low- and high-NLR is lacking and the cutoff values of NLR have been variably reported by using median or a receiver operating characteristics curve methods; mainly 2–3 (range, 1.2 to 5.0) in ESCC [14]. However, rather than the cutoff value itself, the findings that high NLR above a certain level and its increase from baseline have been considerably associated with poor prognosis are important. The role of an on-treatment increase in the NLR as a poor prognostic factor for ICIs was also reported in other cancer types, including melanoma, NSCLC, and renal cell carcinoma [26,27]. Whereas the baseline NLR might indicate the general immune status of a patient, the change in NLR after ICI therapy might indicate the actual process of ICI-induced immune response. The prediction of treatment response to ICI, especially during the very early period, is of interest given the rapid clinical deterioration seen in a considerable number of patients receiving ICIs. Thus, the change in NLR as early as post-cycle 1 could be a valuable clinical tool to predict a systemic immune response in patients treated with ICI. A recent study also suggested that a systemic immune response in patients responding to ICIs can be observed as soon as 14 days after treatment initiation by 18F-fluorodeoxyglucose positron emission tomography, appearing as increased metabolic activity in the spleen [28].
Of note, the current study validated previous findings which showed the negative impact of antibiotic use on the efficacy of ICIs in patients with various cancers including NSCLC, renal cell carcinoma, and melanoma [29,30]. Recently, the gut microbiota has emerged as a key player in cancer immunotherapy via the modulation of the antitumor immune response. The gut microbiota enhance the function of dendritic cells by increasing tumor antigen presentation, cytokine production, recruitment and activation of interferon-γ–producing tumor-antigen-specific effector T cells, and trafficking of CD4+ memory T-cells from mesenteric and draining lymph nodes to the tumor microenvironment, as well as decreasing regulatory T cells and myeloid derived suppressor cells [29,30]. Given that even short-term antibiotics can shift the gut microbiome to long-term alternative dysbiotic states, and their potential harmful effects on cancer immunotherapy, physicians should be judicious with antibiotics use in patients with cancer who are likely to receive ICIs. Although there is concern that patients who recently received antibiotics would have concomitant medical conditions, there were no significant differences in ECOG performance status (≥ 2) (36.8% vs. 45.0%) and hypoalbuminemia (42.1% vs. 39.0%) between patients who received antibiotics and those who did not.
This study has some limitations. First, this study had a limitation in evaluating predictive values as a single-armed design with small number of patients. Second, being a retrospective study, there were missing laboratory values, inducing a risk of potential selection bias. Third, there may be controversies regarding the cutoff values of NLR, PLR, and PNI. Fourth, the evaluation of PD-L1 status was available in only two-thirds of patients with archival tissues and most of them were obtained at the time of diagnosis, not just before ICI administration as 2nd line or ≥ 3 rd line, which could limit the predictive role of PD-L1 status. However, this study has investigated impact of all potential clinical and laboratory factors on the efficacy of ICIs in patients with advanced ESCC.
In conclusion, the current study confirmed the efficacy of ICIs in patients with advanced ESCC after the failure of prior standard chemotherapy in real practice. The treatment outcomes of ICIs may depend on the host immune-nutritional status, and could be predicted using clinical and routine laboratory factors at baseline and early after treatment initiation.
Electronic Supplementary Material
Supplementary materials are available at Cancer Research and Treatment website (https://www.e-crt.org).
Notes
Ethical Statement
The study protocol was approved by the Institutional Review Board of the AMC (study number: 2019-1639) and has been confirmed for waiver of informed consent. This study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice.
Author Contributions
Conceived and designed the analysis: Kim JH, Kim SB, Park SR.
Collected the data: Kim JH, Ahn B, Hong SM, Jung HY, Kim DH, Choi KD, Ahn JY, Kim SB, Lee JH, Na HK, Kim JH, Kim YH, Kim HR, Lee HJ, Park SR.
Contributed data or analysis tools: Kim JH, Kim SB, Park SR.
Performed the analysis: Kim JH, Kim SB, Park SR.
Wrote the paper: Kim JH, Park SR.
Conflict of interest
Conflict of interest relevant to this article was not reported.
Acknowledgments
This research was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF) & funded by the Korean government (MSIT) (No.2019M3E-5D4064636) and grants from National Cancer Centre, Korea (NCC-1911267).