Cisplatin-Based Chemotherapy Is a Strong Risk Factor for Thromboembolic Events in Small-Cell Lung Cancer
Article information
Abstract
Purpose
Cisplatin-associated arterial and venous thromboembolic events (TEEs) are becoming an increasing concern. In patients with small-cell lung cancer (SCLC) who are treated using cisplatin-based chemotherapy, we assume that the overall risk of TEEs is high. However, cisplatin-associated vascular toxicity in patients with SCLC has been overlooked to date. The aim of this study was to determine the incidence of TEEs in patients with SCLC and to analyze the predictors for TEE occurrence.
Materials and Methods
We retrospectively analyzed 277 patients who received chemotherapy for SCLC between 2006 and 2012. As the influence of chemotherapy on TEE occurrence developed after its initiation, a time-dependent Cox regression analysis was used to estimate the significant predictors for TEE.
Results
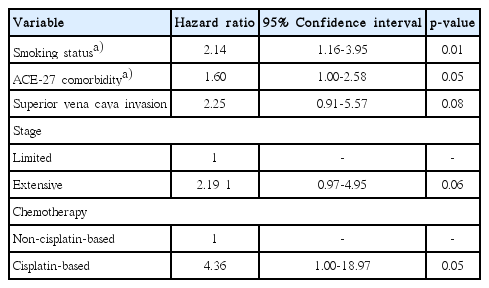
Among the 277 patients, 30 patients (11%) developed a TEE. The 3-month, 6-month, and 1-year cumulative incidences of TEEs were 5.0%, 9.1%, and 10.2%, respectively. Of 30 total TEEs, 22 (73%) occurred between the time of initiation and 4 weeks after the last dose of platinum-based chemotherapy. Approximately 218 patients (79%) received cisplatin-based chemotherapy. In multivariate analysis, cisplatin-based chemotherapy was an independent risk factor for TEE occurrence (hazard ratio [HR], 4.36; p=0.05). Variables including smoking status (common HR, 2.14; p=0.01) and comorbidity index (common HR, 1.60; p=0.05) also showed significant association with TEE occurrence.
Conclusion
The 1-year cumulative incidence of TEE is 10.2% in Asian patients with SCLC. Cisplatin-based chemotherapy in SCLC might be a strong predictor for the risk of TEE.
Introduction
Patients with malignancy have a 4- to 8-fold increased risk of venous thromboembolism compared to those without malignancy [1,2]. The risk of thrombosis varies according to the site of the cancer, tumor burden at diagnosis, and cancer-related treatment [3,4].
Among chemotherapeutic agents, cisplatin in particular has been associated with an increased risk of a thromboembolic event (TEE) [5-8]. In a recent study by Moore et al. [7] including 932 patients with solid malignancies receiving cisplatin-based chemotherapy, 18% of the patients experienced a TEE. Therefore, we can assume that the risk of TEE is considerable in patients with small-cell lung cancer (SCLC); cisplatin in combination with etoposide or irinotecan is the most commonly used regimen [9,10]. However, cisplatin-associated vascular toxicity in patients with SCLC has been overlooked to date. In addition, no studies primarily evaluating the risk of TEE in patients with SCLC have been reported.
The aim of the current study was to investigate the incidence and characteristics of TEE in patients with SCLC who received platinum-based chemotherapy.
Materials and Methods
1. Patient population
All patients who received chemotherapy for SCLC and underwent at least 4 weeks of follow-up after their initial chemotherapy session between January 2006 and June 2012 were analyzed retrospectively. This study was approved by the Institutional Review Board of Seoul National University Hospital (IRB No. H-1102-013-349).
Comorbidity was assessed using the Adult Comorbidity Evaluation 27 (ACE-27), a validated comorbidity instrument used for assessment of patients with cancer [11]. Grades were assigned according to the severity of an individual’s organ decompensation and its prognostic impact. We calculated the Khorana risk score for prediction of chemotherapy-associated thromboembolism using the body mass index (BMI), hemoglobin level, and platelet and white blood cell counts [12]. SCLC stages were classified as either limited or extensive, with the limited-stage confined to the primary tumor and its regional nodal involvement. Treatment characteristics were also collected, including the treatment strategy, chemotherapy regimen, exposure to cisplatin, number of cisplatin cycles, and cumulative cisplatin dose.
2. Identification of TEE
We defined TEE to include arterial thrombosis, deep vein thrombosis (DVT), pulmonary embolism (PE), and thrombosis in other vascular territories except for superficial vein. All reports of radiological (computerized tomography [CT], CT angiography, Doppler ultrasonography, and conventional angiography) or nuclear medicine (ventilation/perfusion scans) studies were reviewed in order to identify a venous or arterial TEE. Two independent investigators (Y.-G.L. and E.L.) double-checked and confirmed the occurrence of TEE by retrospective review of electrical medical records and results of imaging studies. Two or more TEEs occurring either simultaneously or at different times were counted as one event.
3. Statistical analyses
The primary study objective was to determine the cumulative incidence and characteristics of TEEs in patients with SCLC. The time-to-TEE was calculated by taking the difference in time from the date of cancer diagnosis to the first identified date of TEE occurrence. The cumulative hazard estimates for TEE were calculated using the Nelson-Aalen method and compared between groups using log-rank tests. The secondary study objective was to investigate the predictive risk factors for the occurrence of a TEE, which was analyzed using Cox time-dependent covariate regression analysis. When a linear effect of a categorical variable was suspected, we calculated the common hazard ratio (HR) from one group to the next. Variables that were found to be significant with p ≤ 0.05 using a stepwise backward-selection method were entered into a final multivariate model. All analyses were performed using Stata ver. 12.0 (StataCorp, College Station, TX).
Results
1. Baseline and treatment characteristics of the study population
Overall, 306 patients were diagnosed with SCLC during the study period; 277 received chemotherapy and were included in the current study. Twenty-nine patients were excluded; 26 patients did not receive chemotherapy due to old age and/or poor performance status, and three patients were found to have a TEE at the time of cancer diagnosis.
Patients’ baseline and treatment characteristics are summarized in Tables 1 and 2, respectively. The Khorana risk score is included in Table 1. Of the total 277 patients, eight (3%) had experienced cerebrovascular events and 14 (5%) had experienced coronary artery disease including either angina or myocardial infarction. None had experienced both cerebrovascular events and coronary heart disease. With regard to the first-line chemotherapy, 206 patients (74%) received a cisplatin-based regimen and 71 (26%) received a carboplatin-based regimen. Of these 71 patients, 11 (16%) were treated with a cisplatin-based regimen as second-line chemotherapy. In comparison of the baseline characteristics after the first-line chemotherapy, patients receiving an initial carboplatin-based regimen had a poorer performance status (p=0.009) and more severe organ decompensation (p < 0.001) compared to those receiving an initial cisplatin-based regimen.
2. Incidence, timing and characteristics of TEE development
Among the 277 patients included in this study, 30 patients (11%) developed a TEE. The 3-month, 6-month, and 1-year cumulative incidences of TEEs were 5.0%, 9.1%, and 10.2%, respectively. Of a total of 30 TEEs, all TEEs occurred in patients who had received a platinum-based chemotherapy; 22 (73%) occurred between the time of initiation and four weeks after the last dose of platinum-based chemotherapy and the remaining eight (27%) occurred during best supportive care after the end of chemotherapy. Among 22 TEEs associated with platinum-based chemotherapy, 20 (91%) occurred during cisplatin-based chemotherapy and two (9%) occurred during carboplatin-based chemotherapy. Among 20 TEEs related to the cisplatin-based chemotherapy, the majority (90%) occurred during the first-line cisplatin chemotherapy (between the time of initial dose of chemotherapy and 4 weeks after the last dose) and the other 10% occurred thereafter. Fig. 1 shows the cumulative incidence of TEEs after first-line chemotherapy. Overall, the median time from the time of cancer diagnosis until TEE occurrence was 132 days (interquartile range, 58 to 394 days).

Cumulative incidence of thromboembolic events (TEEs) according to chemotherapeutic regimen. HR, hazard ratio.
The majority of events were the occurrence of only PE (n=15), followed by arterial thrombosis (cerebrovascular accidents+central arterial thrombosis, n=7), DVT combined with PE (n=4), and only DVT (n=4).
3. Univariate and multivariate analyses of baseline and treatment characteristics
In univariate analyses, the smoking status (current vs. non-smoker), stage of SCLC, and invasion of the superior vena cava showed association with a significant increase in the risk of TEE (Table 1). Previous history of major vascular events (cerebrovascular events or coronary artery disease) was not associated with TEE occurrence (unpublished data). The study concluded that among the predictive variables included in the Khorana risk model, none showed significant association with TEE occurrence. The overall Khorana risk score was also not predictive of TEE occurrence. In analysis of the data by treatment variables, radiotherapy did not show association with TEE occurrence. Cisplatin-based chemotherapy showed association with increased risk of TEE, which was marginally significant (HR, 3.66; 95% confidence interval [CI], 0.87 to 15.43; p=0.08). However, median dose of cisplatin did not show correlation with the TEE (p=0.20).
In multivariate analysis that included treatment variables as time-dependent covariates, cisplatin-based chemotherapy was an independent risk factor for TEE occurrence (HR, 4.36; 95% CI, 1.00 to 18.97; p=0.05). Variables including the smoking status (common HR, 2.14; 95% CI, 1.16 to 3.95; p=0.01) and ACE-27 comorbidity (common HR, 1.60; 95% CI, 1.00 to 2.58; p=0.05) also showed significant association with TEE occurrence (Table 3).
Discussion
This is the first study to focus primarily on SCLC and to evaluate the risk of TEE in patients with SCLC. In our retrospective analysis of 277 patients treated with platinum-based chemotherapy for SCLC, the 3-month, 6-month, and 1-year cumulative incidences of TEEs were 5.0%, 9.1%, and 10.2%, respectively. In multivariate analysis, the current smoking status, higher comorbidity index, and cisplatin-based chemotherapy were independent predictors for increased risk of TEE. Compared to patients receiving non-cisplatin based chemotherapy, risk of TEE was 4.4 times higher for patients receiving cisplatin-based chemotherapy.
In a population-based study including 13,542 patients with SCLC conducted in California between 1993 and 1999, the 2-year incidence of venous thromboembolism in patients with SCLC was 2.5% [13]. Given the low incidence reported in the Western epidemiologic study, our result showing a 1-year cumulative incidence of 10.2% seems high. However, in recent studies focusing on the relationship between cisplatin and thromboembolism, the incidence of TEE was 8.4% to 12.9% in patients primarily receiving cisplatin-containing chemotherapy [5,14]. A recent retrospective study analyzing 932 patients treated with cisplatin-based chemotherapy for various cancers also reported a similar incidence of TEEs (11.8%) in 204 patients with lung cancer [7]. In our study, almost 80% of patients were exposed to cisplatin-based chemotherapy. In addition, the risk of TEE in patients receiving cisplatin-based chemotherapy was 4.4 times higher than that of those receiving non-cisplatin based chemotherapy. The results of the current study support the hypothesis that cisplatin-based chemotherapy was indeed a major contributor to the high incidence rates of TEE.
Despite clinical evidence of cisplatin-induced hypercoagulability, the pathophysiology remains unclear. Several studies have suggested that cisplatin may cause the following actions: endothelial damage via induction of procoagulant microparticles, an increase in the procoagulant activity of red blood cells, and modulation of tissue factor on human monocytes. These characteristics may contribute to cisplatin-induced hypercoagulability [15-18].
In terms of dose-response relationship, according to our data, the cumulative dose of cisplatin did not show correlation with TEE occurrence. This finding is consistent with results from a previous large retrospective study by Moore et al. [7], however, a recent meta-analysis suggested that the higher dose of cisplatin was associated with the heightened risk of thromboembolism [8]. These conflicting results can be explained in part by the study design (retrospective vs. prospective) and the size of the study. Therefore, conduct of well-designed and prospective studies is warranted.
In contrast to the well-known risk factors (smoking status, comorbidity index, and cisplatin-based chemotherapy) for thrombosis in our study, the Khorana score was not predictive of development of TEE. This finding may be explained by the fact that the overall risks for TEE in patients receiving cisplatin-containing chemotherapy were much higher compared to those in the study population included in the Khorana study [12]. The very low proportion of patients with a BMI ≥ 35 kg/m2 in this study in comparison with the Khorana study (0.4% vs. 12.3%) also contributes to the lack of predictive significance of the Khorana variables. Given the difference in cut-off points of BMI as an indicator of obesity between Asians and Caucasians, an area-specific cut-off point for BMI among the Khorana variables should be taken into consideration.
Our results are limited by selection bias due to the retrospective nature of the study. The exclusion of patients who did not receive chemotherapy because of poor performance status and/or severe organ decompensation may lead to an underestimation of the real burden of TEE in patients with SCLC. Since the total number of patients with SCLC and TEE is low due to their rare incidence, our analyses were most likely underpowered for detection of statistical significance. Therefore, conduct of additional studies with large sample sizes will be necessary in order to confirm our findings.
Conclusion
In conclusion, in the current study, the 3-month, 6-month, and 1-year cumulative incidences of TEEs in Asian patients with SCLC were 5.0%, 9.1%, and 10.2%, respectively. Cisplatin-based chemotherapy in SCLC might be a strong predictor for the risk of TEE.
Notes
Conflict of interest relevant to this article was not reported.
Acknowledgements
This study was supported by grant No. 0720142084 from the National Research Foundation of Korea and by a grant of Seoul National University College of Medicine (800-20140171). The authors wish to thank Leo Song (University College Dublin School of Medicine, Dublin, Ireland) for the assistance in data collection.