AbstractPurposeHistone deacetylase inhibitors (HDACis) are epigenetic regulators and used clinically for hematopoietic malignancies. Recently, HDACis have received attention as a factor that modulates the immune system. In this study, the role of histone deacetylase (HDAC) expression as a predictive marker in lung cancer patients who were treated with immune checkpoint inhibitors (ICIs) and the role of HDACi and ICI combination treatment in the mouse tumor model were analyzed.
Materials and MethodsThe overall response rate (ORR) and progression-free survival (PFS) were analyzed by the expression of HDAC. In vitro assay, the mRNA and protein expression levels of cytokines and programmed death-ligand 1 (PD-L1) were analyzed after HDACi treatment. In vivo assay, TC-1 tumor-bearing mice were treated with HDACi and mouse programmed cell death 1 (PD-1) inhibitor.
ResultsThe HDAC6 low expression group showed high ORR and prolonged PFS. When the selective HDAC6 inhibitor was administered to the A549 cell line, the levels of interleukin-1β and interleukin-6 decreased and the expression of PD-L1 was reduced. Mice that received both the mouse PD-1 inhibitor and pan-HDACi had a smaller tumor size than that of the mice from the control group. Moreover, mice treated with the mouse PD-1 inhibitor and pan-HDACi generated greater numbers of E7-specific CD8+ T cells.
ConclusionHDAC6 expression can predict the prognosis of non–small cell lung cancer patients who were treated with ICIs. Furthermore, co-treatment with HDACi and PD-1 inhibitor was shown to decrease the tumor growth rate and create a favorable tumor microenvironment for cytotoxic T lymphocytes in the TC-1 mouse model.
IntroductionThe therapy for non–small cell lung cancer (NSCLC) has entered a new era with the emergence of immune checkpoint inhibitors (ICIs). ICIs attack cancer cells by activating cytotoxic T lymphocytes (CTLs) that have been inhibited by the immune checkpoint [1]. Compared to cytotoxic chemotherapy, ICIs are ideal cancer treatment with a long duration of efficacy and few side effects [2]. However, the tumor cells alter the tumor microenvironment (TME) to create a favorable environment for the cancer cells, thus enabling immune escape [3]. Cancer cells can inhibit CTLs by recruiting myeloid-derived suppressor cells (MDSCs) or regulatory T cells (Tregs) in the TME [4,5] and by secreting various cytokines [6]. Because of these immunosuppressive immune cells and cytokines, antigen sensitization is lowered, and the migration, infiltration, and activation of CTLs are suppressed [7]. Therefore, the use of ICIs for treating NSCLC is limited due to treatment response rates as low as 15%–25%. An effective biomarker that can predict the treatment response to ICIs is highly desired, as are drugs that can enhance the therapeutic effect of ICIs by creating a favorable TME for CTLs.
Histone deacetylase (HDAC) and histone deacetylase inhibitors (HDACis) are attracting attention as factors that can predict the treatment response to ICIs and increase their efficacy [8]. HDAC and HDACi are known as factors involved in cell proliferation, differentiation, and apoptosis by regulating gene expression [9]. In addition, HDACi can modulate the immune system and chronic inflammation [10]. When the HDAC expression is increased, a chronic inflammatory condition can occur, which can create a favorable environment for cancer [11]. This study aimed to analyze the correlation between HDAC expression and the prognosis of NSCLC patients and the treatment prognosis of ICIs. In addition, the effects of HDACi on the TME were analyzed through in vitro and mouse experiments, and the effect of combined treatment with ICI was to be evaluated.
Materials and MethodsThe study was divided into three parts. First, the prognosis of patients treated with ICIs was analyzed through immunohistochemistry (IHC) of HDAC (Correlation between HDAC expression and ICI treatment outcome). Second, changes in the TME were assessed by HDACi treatment in a lung cancer cell line (effects of HDACi on tumor microenvironment). Third, changes in the TME were examined by HDACi treatment in the TC-1 mouse model (combined effect of ICI and HDACi in mouse tumor model).
1. Study design and participantsForty-six NSCLC patients who received ICIs at tertiary academic Hospitals were retrospectively analyzed from December 1, 2016 to June 30, 2019. Based on the 8th edition of the tumor, node and metastasis (TNM) staging, NSCLC patients with stage IIIA or higher and relapsed NSCLC patients were included. Patients over the age of 18 that had an Eastern Cooperative Oncology Group performance status of less than level 2 were included. Patients were treated with either pembrolizumab, atezolizumab, or nivolumab. Pembrolizumab and atezolizumab were administered for 3 weeks on a 1-cycle basis, and nivolumab was administered for 2 weeks on a 1-cycle basis. ICI treatment continued until patients could no longer be treated due to disease progression or side effects. Responses were evaluated through chest computed tomography (CT) every two cycles for pembrolizumab and atezolizumab patients and every three cycles for nivolumab patients. Positron emission tomography CT was also performed when needed for response evaluation. Responses were evaluated based on Response Evaluation Criteria in Solid Tumors ver. 1.1. Depending on the expression level of HDAC, the overall response rate (ORR), progression-free survival (PFS), and overall survival (OS) were analyzed. ORR is defined as the complete response or partial response based on the best response. In cases where the condition was judged to be a progressive disease (PD) as determined by CT, treatment was continued when it was estimated to be a pseudo-progression according to the judgment of the clinician. PFS was calculated from the start of ICIs treatment to the date of discontinuation; OS was calculated from the start of ICIs treatment to the date of death. Patients who used ICIs in the fourth or higher lines, patients for whom the response could not be evaluated, or patients who did not complete the second cycle were excluded.
2. Immunohistochemical stainingIHC was performed using initially diagnosed samples of NSCLC patients who received ICI. HDAC1 and HDAC6 were selected as candidate members from the HDAC Class I and HDAC Class II families, respectively. Rabbit monoclonal antibodies against HDAC 1 (anti-HDAC1 antibody [EPR460(2)] ab109411, Abcam, Cambridge, MA) and 6 (anti-HDAC6 antibody [EPR1698(2)] ab133493, Abcam) were used. HDAC expression was evaluated in the same way as programmed death-ligand 1 (PD-L1) expression was analyzed. Two pathologists and two respiratory specialists evaluated HDAC expression levels in a blinded manner, and complete observer agreement was conducted. The score was recorded as a percentage of tumor cells that showed any intensity of cytoplasmic immunoreactivity. We classified the examined cases into two groups according to their IHC scores as follows: HDAC-high group (HDAC expression of over 51%) and HDAC-Low group (HDAC expression of 0%–50%) (Fig. 1).
3. Cell lines and reagentsThe A549 cell line, a human lung carcinoma cell line, was purchased from the Korean Cell Line Bank (Seoul, Korea) and grown in Roswell Park Memorial Institute 1640 media (Welgene, Gyeongsan, Korea) containing 10% fetal bovine serum as a growth factor and 100 mg/mL penicillin/streptomycin as antibiotics. Cells were plated at 0.3×106 cells/well on 6-well plates. Treatments were administered in consideration of the half maximal inhibitory concentration (IC50). Cells were incubated at 37°C with a 5% CO2 atmosphere in an incubator. Cells were harvested 72 hours after treatment. The mouse lung tumor line TC-1, which was transduced to express the E7 oncogene of HPV-16, was provided by T. C. Wu [12]. TC-1 cells were grown in RPMI medium 1640 supplement with 10% fetal bovine serum, 50 U/mL penicillin/streptomycin, 2 mM L-glutamine, 1 mM sodium pyruvate, and 2 mM non-essential amino acids and 2-mercaptoethanol at 37 °C in a 5% CO2 atmosphere. Nexturastat A (molecular weight=341.4, catalog No. S7473) was selected from among the HDACi, as it is a potent and selective HDAC6 inhibitor with an IC50 of 5 nM and > 190-fold selectivity over other HDACs. Nexturastat A was purchased from Selleckchem (Houston, TX). New Pan-HDAC inhibitor, M166 was supplied by Chong Kun Dang Pharm.
4. RNA isolation and reverse transcription polymerase chain reactionTotal RNA was isolated from cells using TRIzol solution (Life Technologies, Carlsbad, CA). Complementary DNA was prepared using a CycleScript Reverse Transcription PreMix (dT20) kit (Bioneer, Daejeon, Korea). Transcripts were amplified using Taq PCR Master Mix reagent (Bioneer). Polymerase chain reaction (PCR) was performed under the following conditions: pre-denaturation at 95°C for 1 minute followed by cycles of denaturation at 95°C for 30 seconds, annealing at 60°C for 30 seconds, and polymerization at 72°C for 30 seconds; 30 and 25 cycles were used for the amplified genes and glyceraldehyde 3-phosphate dehydrogenase (GAPDH), respectively. The final extension step was at 72°C for 5 minutes, and the hold step was at 4°C. PCR primers of human GAPDH, human interleukin (IL)-IL-1β, IL-6, and PD-L1 are denoted in S1 Table.
5. Animal modelFemale C57BL/6 mice (5–6 weeks old) were purchased from Orient Bio Animal Inc. (Seoul, Korea). For the in vivo tumor treatment, 2×105 TC-1 tumor cells per mouse were subcutaneously injected into the left or right flank area of 6–7-week-old C57BL/6 mice. After 10–12 days, similar size tumor-bearing mice were divided into four groups (five per group), each receiving a different treatment regimen: group 1 received no treatment after the TC-1 tumor challenge, group 2 was treated with anti-mouse programmed cell death 1 (PD-1) antibodies (CD279) (catalog No. RMP1–14, Bio X Cell, Lebanon, NH) by intraperitoneal injection (200 μg/mouse) 3 times per week for 3 weeks, group 3 was treated with new pan-HDACi (M166) by intraperitoneal injection (based on 50% and 60% of IC50) twice per week for 3 weeks, and group 4 was treated with both anti-mouse PD-1 antibodies (CD279) and new pan-HDACi (M166). The new pan-HDACi (M166) was injected two times per week by M166 drug toxicity. The mice were monitored thrice a week by examination and palpation. Tumor size was monitored by measuring the longest dimension (length) and the shortest dimension (width) of the tumors using dial calipers at 3-day intervals. Tumor volume was calculated by the following formula: tumor diameter= length×(width×width)×0.5.
6. Intracellular cytokine staining and flow cytometry analysisPooled splenocytes from tumor-bearing mice that were treated with the various treatment regimens were harvested 7 days after the last treatment and incubated for 24 hours with 1 μg/mL of the E7 peptide containing an MHC class I epitope (amino acids 49–57, RAHYNIVTF) in the presence of GolgiPlug (BD Pharmingen, Franklin Lakes, NJ). The stimulated splenocytes were then washed once with fluorescence-activated cell sorting (FACS) buffer and stained with PE-conjugated monoclonal rat anti-mouse CD8a antibodies (clone 53.6.7). Intracellular interferon γ (IFN-γ) was stained with FITC-conjugated rat anti-mouse IFN-γ. All antibodies were purchased from BD Pharmingen. Flow cytometry analysis was performed using a FACS Fortessa instrument with Flow Jo software (BD Biosciences, San Jose, CA).
7. Statistical analysisData were analyzed using SPSS ver. 20 software (IBM Corp., Armonk, NY). Continuous variables were reported as the means±standard deviations, and categorical variables were reported as a number and percentage. Continuous variables were compared using Student’s t test. Categorical variables were compared using the chi-square test or with Fisher’s exact test, which was used when the expected number of events was < 5. The Kaplan-Meier method and log-rank test were used for analyzing PFS and OS. p-values < 0.05 were considered statistically significant.
Results1. Correlation between HDAC6 expression and the effect of ICI treatment in NSCLC patients treated with ICIRegarding the expression of HDAC1 in lung cancer tissues, 41 patients had 100% expression, and 45 patients showed more than 50% expression. Overall, HDAC1 primarily exhibited high expression; thus, further analysis could not be performed. However, for the expression of HDAC6, there were 11 people in the HDAC6-Low expression group and 35 people in the HDAC6-High expression group. These two groups, which were classified according to HDAC6 expression level, were compared (Fig. 2). The mean age was 69.8±8.8 years, and about 80% of patients were male. Moreover, 30 patients had ex- or current smoking history, 31 had squamous cell carcinoma, 34 had stage IV cancer, and pembrolizumab was administered the most frequently. The baseline characteristics revealed no statistical differences between the two groups (Table 1).
There were no patients who showed complete response in either group. In the first response evaluation after two cycles, five patients had PD in the HDAC6 high group, whereas no patients had PD in the HDAC6-Low expression group. The ORR showed a statistically significant difference in both groups (p=0.042) (Table 2). The waterfall plot analysis indicated that the overall size of the tumor mass decreased more in the HDAC6-Low group. On average, tumor size in the HDAC6-High expression group decreased by 13.4%±35.8%, while that in the HDAC6-Low expression group decreased by 38.6%±31.8% (Fig. 3).
The average follow-up period for the HDAC6-High group was 417.1±158.2 days, and that for the HDAC6-Low group was 506.9±182.3 days. During the follow-up period, 26 patients showed PD in the HDAC6-High group, and only four patients in the HDAC6-Low group showed PD (p=0.035). Furthermore, 15 and three patients died in the HDAC6-High expression and HDAC6-Low expression groups, respectively (p=0.090) (Fig. 4).
2. HDACi decreased inflammatory cytokine and PD-L1 expressionTo find the effect of HDAC6 at the molecular level in lung cancer, the effect of HDAC6 inhibitor on inflammatory markers was evaluated. Nexturastat A (HDAC6 inhibitor) treatment decreases the IL-1β and IL-6 inflammatory cytokine mRNA expression levels (Fig. 5A). It was confirmed that the expression levels of IL-1β and IL-6 were the lowest following treatment with Nexturastat A at a concentration that was 1.5 times its IC50. Although inflammatory cytokine levels were increased following treatment with Nexturastat A at concentrations up to 2.0-fold that of its IC50, they were still lower than those of the control (no treatment) group. Especially, IL-1β cytokine as a pulmonary inflammatory marker, expression levels were dose-dependently decreased by Nexturastat A (Fig. 5B). These experimental results are performed three independently. These figures are the representative result and band intensity measured by the Image J program, then average and standard deviation are calculated by Excel program. These results indicated that Nexturastat A is efficiently reduced proinflammatory cytokines of IL-1β and IL-6.
Another effect of Nexturastat A, when treated with Nexturastat A, the mRNA expression levels of PD-L1 were reduced. Additionally, this change followed a dose-dependent pattern (Fig. 6A). Also, PD-L1 protein expression levels were dose-dependently decreased by Nexturastat A (Fig. 6B). New pan-HDACi (M166) also decreased PD-L1 protein expression, dose-dependently (Fig. 6B). These results indicated that Nexturastat A efficiently reduced PD-L1 mRNA and protein expression. Also, new pan-HDACi (M166) efficiently reduced PD-L1 protein expression. These meanings indicated that Nexturastat A and new pan-HDACi (M166) efficiently reduced the expression of PD-L1 cancer cell surface protein.
3. Combination treatment of HDACi and PD-1 inhibitor generates anti-tumor effects and elicits E7-specific CD8+ T cells in TC-1 tumor-bearing miceTo determine whether the reduction in the expression of inflammatory cytokines (IL-1β, IL-6) and PD-L1 influences tumor growth, we made a TC-1 tumor-bearing mice model (Fig. 7A). Mice that received both the mouse PD-1 inhibitor (CD279) and new pan-HDACi (M166) had a smaller tumor size compared to that of the control group (p=0.03). There were no statistically significant differences between the ICIs and new pan-HDACi (M166) groups (Fig. 7B). These results indicated that new pan-HDACi (M166) was synergistic effect of ICI function.
In addition, mice receiving both mouse PD-1 inhibitor and new pan-HDACi (M166) generated more significantly E7-specific IFN-γ–producing CD8+ T cells. Although the difference was little, more E7-specific IFN-γ–producing CD8+ T cells were produced by the new pan-HDACi (M166) treatment group than by the mouse PD-1 inhibitor group (Fig. 7C). These results indicated that E7-specific IFN-γ–producing CD8+ T cells were more expression by ICI co-treat with new pan-HDACi (M166). These results reflex the synergistic effect of ICI function by new pan-HDACi (M166) through an immune-mediated mechanism.
DiscussionThis research has demonstrated that HDAC6 expression can predict responsiveness to ICIs. The high HDAC6 expression indicates that the prognosis of the patient is poor. Besides, selective HDAC6 inhibitors (Nexturastat A) have been shown to create a favorable environment for CTLs. Selective HDAC6 inhibitors (Nexturastat A) can lower inflammatory cytokine secretion, regulate PD-L1 expression, and alter the composition of CTLs.
NSCLC has one of the highest mortality rates among many solid cancers. In advanced non-small cell carcinoma, the life expectancy is approximately 1 year even with cytotoxic chemotherapy. As a new treatment to overcome this challenge, ICIs have been developed and are actively used in real clinical fields. However, cancers evade immune mechanisms by regulating the TME. Because of the various mechanisms of immune escape, ICIs have shown a lower therapeutic response rate [13]. To overcome these limitations, it is necessary to develop effective biomarkers that can predict the therapeutic response to ICIs [14]. Moreover, there is a need for research on drugs that can increase the response rate by altering the TME to favor ICIs [15].
Research on various cytokines and cells that can influence the TME is actively underway [16]. Chronic inflammatory conditions have been linked to the development, progression, and exacerbation of cancer and have been found to inhibit the effectiveness of ICIs [17]. IL-1β and IL-6 have been identified as representative inflammatory cytokines that can inhibit the effects of ICIs. IL-1β can promote cancer by controlling angiogenesis and endothelial cells around cancer cells and can directly suppress the immune system using MDSCs [18]. IL-1β blockers (anakinra and canakinumab) have also been examined in clinical studies [19]. Moreover, IL-6 has been shown to inhibit the invasion of CTLs into the tumor mass and suppress ICIs by activating MDSCs and Tregs [20]. Drugs capable of inhibiting IL-1β and IL-6 are thought to increase the efficacy of ICIs.
HDACi inhibits the exocytosis of inflammatory cytokines and the synthesis of inflammation-related substances (nitric oxide and prostaglandin E2) [21]. Currently, HDACi is one type of drug that is being studied in combination therapy with ICIs [22,23]. HDACi is well known as a drug type that regulates various cell functions, such as cell proliferation, differentiation, and apoptosis, by regulating gene transcription [24]. It has recently been found that HDACi can regulate inflammation, infection, and immunity through mechanisms that are independent of histones. Moreover, HDACis are thought to be effective in increasing the efficacy of ICIs by creating a favorable environment for CTLs. The currently reported mechanisms underlying their action include increa-sed expression of major histocompatibility complex class, increased levels of cytokines favorable to the activity of CTLs, increased survival of activated CTLs, and decreased levels of MDSCs or Treg cells [25,26].
HDAC has several classes, and their functions vary depending on the class [27]. HDAC class I (HDAC 1, 2, 3, 8) mainly exists in the cell nucleus and regulates gene transcription. The function through which HDACi creates a favorable environment for ICIs is thought to occur in the cytoplasm and not in a region that regulates the chromatin environment. The HDAC class IIB (HDAC 6, 10) is mostly distributed in the cytoplasm. Among these, HDAC6 inhibitors are thought to be able to alter the TME through interactions with non-histone substrates [28]. The Nexturastat A, one of the selective HDAC6 inhibitors, increases the memory capacity of CTLs, and inhibits the activity of pro-tumorigenic M2 macrophages and Treg cells to create a favorable environment for ICIs [25,26]. In this study, Nexturastat A created a favorable environment for CTLs by inhibiting the secretion of inflammatory cytokines such as IL-1β and IL-6.
Research for predicting the prognosis of patients with various solid cancers using HDAC as a marker has long been conducted [29]. However, this study is the first to analyze the prognosis of ICI treatment in NSCLC based on HDAC6 expression. Unlike the expression level of PD-L1, the pathologic criteria for evaluating the expression level of HDAC are yet to be established. When criteria for HDAC expression are established and combined with other biomarkers, a more accurate prognostic analysis will be possible. In some reports, there is a positive correlation between HDAC expression and PD-L1 expression. However, in our clinical data, there was no correlation between HDAC expression and PD-L1 expression. This reflects that different mechanisms may act between HDAC and PD-L1 therapeutics. Although HDACis reduce the expression of PD-L1, their main action is to change the immune status in the TME by influencing immune-suppressive inflammatory cytokines rather than inhibiting PD-L1. This indicates a synergistic effect of HDACis on PD-L1 inhibitors. Among general lung cancer patients, PD-L1 high accounts for about 30%; whereas in our study, 70% of patients had PD-L1 high. The higher proportion of PD-L1 in our study could place a limitation on the analysis of the correlation between HDAC expression and PD-L1 expression. The effects of HDACi on PD-L1 expression and the mechanisms underlying these effects vary depending on the class of HDAC targeted by the HDACi and the type of HDACi. Even when using the same HDACi, the effect differs according to the cell type. For example, SAHA (a representative pan-HDACi) lowered PD-L1 expression in a lung cancer cell line, whereas it increased the PD-L1 expression in a B-cell lymphoma cell line [30].
In cancer treatment, HDACi has been noted only for altering gene transcription. However, HDACi is thought to be a drug type capable of altering the TME through interactions with non-histone substrates. Here, this potential was demonstrated through experiments involving IHC analysis of lung cancer cell lines and mice. HDAC6 low expression group showed good ICI response in NSCLC patients. HDACi modulates inflammatory cytokine and PD-L1 expression to create a favorable environment for ICIs in lung cancer cell line. Combination treatment of HDACi and PD-1 inhibitor generates anti-tumor effects in TC-1 tumor-bearing mice. In the future, clinical trials of the combination therapy of HDACi and ICIs should be actively considered.
Electronic Supplementary MaterialSupplementary materials are available at Cancer Research and Treatment website (https://www.e-crt.org).
NotesEthical Statement This research was approved by the Institutional Review Board (IRB) committee of Korea University Guro Hospital (Approval number: 2019GR0209), which waived the requirement for informed consent due to the retrospective study design. All animal procedures were performed according to approved protocols Institutional Animal Care and Use Committee (IACUC) and in accordance with recommendations for the proper use and care of laboratory animals. AcknowledgmentsThis study was supported by Korean Association for the Study of Targeted Therapy (KASTT) affiliated with Korean Association for Lung cancer (KASTT-201901). The mouse tumor line TC-1 was provided by Dr. T.C. Wu.
Fig. 1Representative tumor microarray core stained with histone deacetylase 6 (HDAC6). The score was recorded as a percentage of tumor cells that showed any intensity of cytoplasmic immunoreactivity. (A) HDAC6-High group (HDAC expression 51%-100%). (B) HDAC6-Low group (HDAC expression 0%-50%). 
Fig. 2Patient flow chart. In total, 92 patients received immune checkpoint inhibitors; 46 patients were excluded based on the exclusion criteria. There were 11 and 35 patients in the histone deacetylase 6 (HDAC6)-Low and HDAC6-High groups, respectively. ECOG PS, Eastern Cooperative Oncology Group performance status. 
Fig. 3Waterfall plot for the overall objective response rate in patients with metastatic non–small cell lung cancer who received immune checkpoint inhibitors. The overall tumor size showed a greater decrease in the histone deacetylase 6 (HDAC6)–Low group. On average, the tumor size of the HDAC6-High group decreased by 13.4%±35.8%, and that of the HDAC6-Low group decreased by 38.6%±31.8%. PD, progressive disease; PR, partial response. 
Fig. 4Kaplan-Meier survival curves for progression-free survival (A) and overall survival (B). (A) Twenty-six patients showed progressive disease (PD) in the histone deacetylase 6 (HDAC6)–High group, whereas only four patients in the HDAC6-Low group showed PD (p=0.035). (B) Fifteen and three patients died in the HDAC6-High and HDAC6-Low groups, respectively (p=0.090). 
Fig. 5Reverse transcription polymerase chain reaction (RT-PCR) and Western blotting analysis of inflammatory cytokines. (A) A549 cells were treated in vitro with Nexturastat A dose-dependently, then mRNA levels of interleukin (IL)-1β and IL-6 were measured by RT-PCR. (B) Protein expression levels of IL-1β and IL-6 in A549 cells were measured by Western blot experiment after treatment with Nexturastat A. GAPDH, glyceraldehyde 3-phosphate dehydrogenase. 
Fig. 6Reverse transcription polymerase chain reaction (RT-PCR) and Western blotting analysis of programmed death-ligand 1 (PD-L1). (A) A549 cells were treated in vitro with Nexturastat A dose-dependently, then mRNA levels of PD-L1 were measured by RT-PCR. (B) Protein expression levels of PD-L1 in A549 cells measured by Western blot experiment after treatment with Nexturastat A or pan-HDACi (M166). GAPDH, glyceraldehyde 3-phosphate dehydrogenase; HDACi, histone deacetylase inhibitor. 
Fig. 7The TC-1 mouse model study. (A) Schematic representation of the anti-mouse programmed cell death 1 (PD-1) antibody (CD279) and new pan-HDACi (M166) treatment regimens. C57BL/6 mice (five per group) were subcutaneously injected with 2×105 TC-1 cells per mouse on day 0. Tumor-bearing mice were treated with CD279 and/or M166 beginning on day 10, as indicated in the timeline. CD279 (200 μg/mouse) was administered at intervals of 2–3 days. M166 was administered via intraperitoneal injection at a dose of 60 mg/kg body weight. (B) Tumor volumes of mice treated with PD-1 antibody and new pan-HDACi (M166). Mice that received both mouse PD-1 inhibitor and new pan-HDACi (M166) had a smaller tumor size compared to that of mice from the control group (p=0.03). (C) Characterization of E7-specific CD8+ T cells. Mice receiving both the mouse PD-1 inhibitor and pan-HDACi generated significantly more E7-specific interferon-γ–producing CD8+ T cells. HDACi, histone deacetylase inhibitors; ICI, immune check point inhibitor; IFN-γ, intracellular interferon γ. 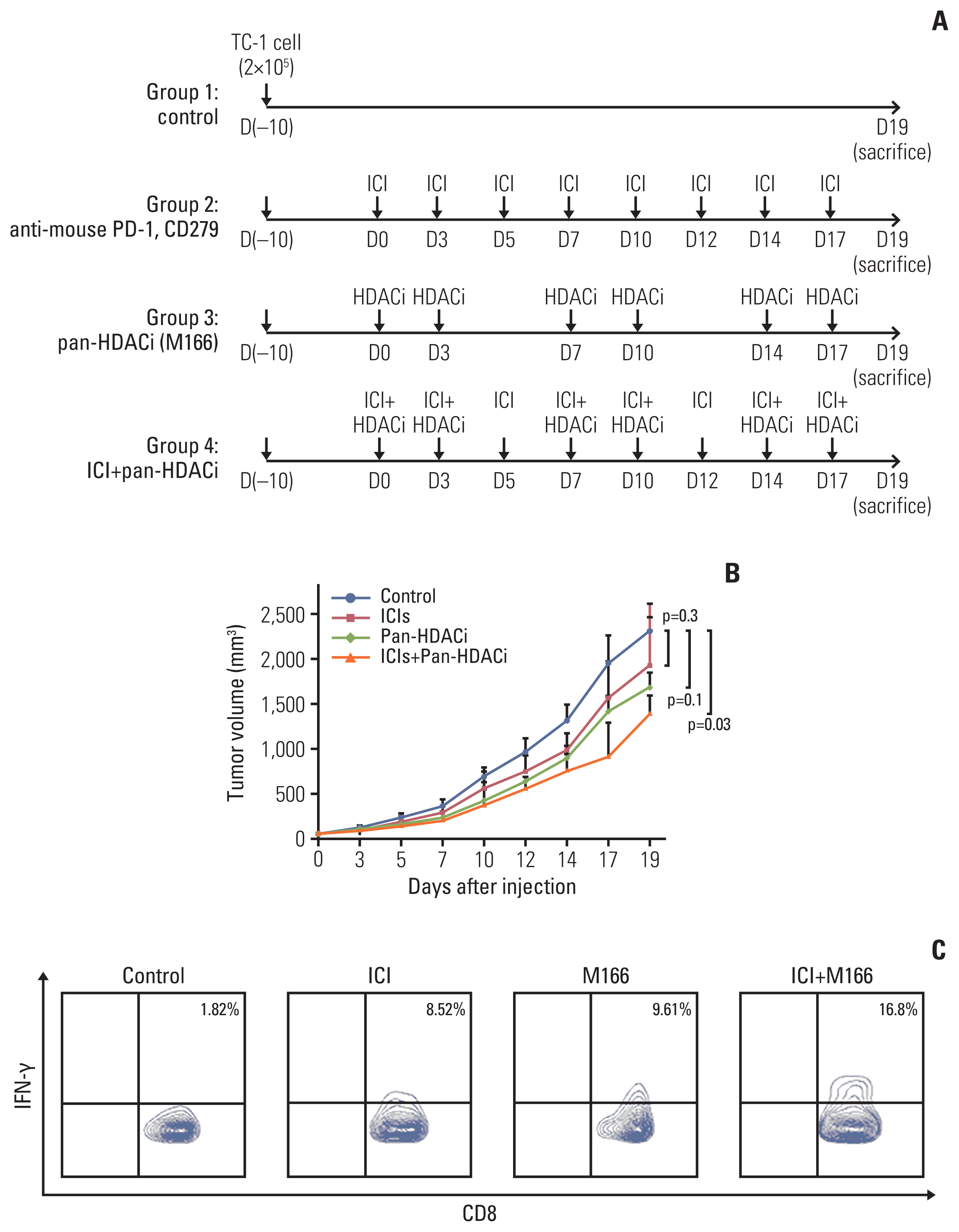
Table 1Baseline characteristics References1. Boussiotis VA. Molecular and biochemical aspects of the PD-1 checkpoint pathway. N Engl J Med. 2016;375:1767–78.
2. Postow MA, Sidlow R, Hellmann MD. Immune-related adverse events associated with immune checkpoint blockade. N Engl J Med. 2018;378:158–68.
3. Spranger S. Mechanisms of tumor escape in the context of the T-cell-inflamed and the non-T-cell-inflamed tumor microenvironment. Int Immunol. 2016;28:383–91.
4. Law AM, Valdes-Mora F, Gallego-Ortega D. Myeloid-derived suppressor cells as a therapeutic target for cancer. Cells. 2020;9:561.
5. Chaudhary B, Elkord E. Regulatory T cells in the tumor microenvironment and cancer progression: role and therapeutic targeting. Vaccines (Basel). 2016;4:28.
6. Bretz NP, Ridinger J, Rupp AK, Rimbach K, Keller S, Rupp C, et al. Body fluid exosomes promote secretion of inflammatory cytokines in monocytic cells via Toll-like receptor signaling. J Biol Chem. 2013;288:36691–702.
7. Messerschmidt JL, Prendergast GC, Messerschmidt GL. How cancers escape immune destruction and mechanisms of action for the new significantly active immune therapies: helping nonimmunologists decipher recent advances. Oncologist. 2016;21:233–43.
8. Wang X, Waschke BC, Woolaver RA, Chen Z, Zhang G, Piscopio AD, et al. Histone deacetylase inhibition sensitizes PD1 blockade-resistant B-cell lymphomas. Cancer Immunol Res. 2019;7:1318–31.
9. Gryder BE, Sodji QH, Oyelere AK. Targeted cancer therapy: giving histone deacetylase inhibitors all they need to succeed. Future Med Chem. 2012;4:505–24.
10. Hull EE, Montgomery MR, Leyva KJ. HDAC Inhibitors as epigenetic regulators of the immune system: impacts on cancer therapy and inflammatory diseases. Biomed Res Int. 2016;2016:8797206
11. Taniguchi K, Karin M. NF-kappaB, inflammation, immunity and cancer: coming of age. Nat Rev Immunol. 2018;18:309–24.
12. Lin KY, Guarnieri FG, Staveley-O’Carroll KF, Levitsky HI, August JT, Pardoll DM, et al. Treatment of established tumors with a novel vaccine that enhances major histocompatibility class II presentation of tumor antigen. Cancer Res. 1996;56:21–6.
13. Reck M, Rodriguez-Abreu D, Robinson AG, Hui R, Csoszi T, Fulop A, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. 2016;375:1823–33.
14. Grigg C, Rizvi NA. PD-L1 biomarker testing for non-small cell lung cancer: truth or fiction? J Immunother Cancer. 2016;4:48.
15. Popovic A, Jaffee EM, Zaidi N. Emerging strategies for combination checkpoint modulators in cancer immunotherapy. J Clin Invest. 2018;128:3209–18.
16. Gabrilovich DI, Ostrand-Rosenberg S, Bronte V. Coordinated regulation of myeloid cells by tumours. Nat Rev Immunol. 2012;12:253–68.
17. Wang D, DuBois RN. Immunosuppression associated with chronic inflammation in the tumor microenvironment. Carcinogenesis. 2015;36:1085–93.
18. Mantovani A, Barajon I, Garlanda C. IL-1 and IL-1 regulatory pathways in cancer progression and therapy. Immunol Rev. 2018;281:57–61.
19. Schenk KM, Reuss JE, Choquette K, Spira AI. A review of canakinumab and its therapeutic potential for non-small cell lung cancer. Anticancer Drugs. 2019;30:879–85.
20. Li J, Xu J, Yan X, Jin K, Li W, Zhang R. Targeting interleukin-6 (IL-6) sensitizes anti-PD-L1 treatment in a colorectal cancer preclinical model. Med Sci Monit. 2018;24:5501–8.
21. Carta S, Tassi S, Semino C, Fossati G, Mascagni P, Dinarello CA, et al. Histone deacetylase inhibitors prevent exocytosis of interleukin-1beta-containing secretory lysosomes: role of microtubules. Blood. 2006;108:1618–26.
22. Tang J, Pearce L, O’Donnell-Tormey J, Hubbard-Lucey VM. Trends in the global immuno-oncology landscape. Nat Rev Drug Discov. 2018;17:783–4.
23. Hontecillas-Prieto L, Flores-Campos R, Silver A, de Alava E, Hajji N, Garcia-Dominguez DJ. Synergistic enhancement of cancer therapy using HDAC inhibitors: opportunity for clinical trials. Front Genet. 2020;11:578011.
24. Li Y, Seto E. HDACs and HDAC inhibitors in cancer development and therapy. Cold Spring Harb Perspect Med. 2016;6:a026831.
25. Knox T, Sahakian E, Banik D, Hadley M, Palmer E, Noonepalle S, et al. Selective HDAC6 inhibitors improve anti-PD-1 immune checkpoint blockade therapy by decreasing the anti-inflammatory phenotype of macrophages and down-regulation of immunosuppressive proteins in tumor cells. Sci Rep. 2019;9:6136.
26. Laino AS, Betts BC, Veerapathran A, Dolgalev I, Sarnaik A, Quayle SN, et al. HDAC6 selective inhibition of melanoma patient T-cells augments anti-tumor characteristics. J Immunother Cancer. 2019;7:33.
27. Banik D, Moufarrij S, Villagra A. Immunoepigenetics combination therapies: an overview of the role of HDACs in cancer immunotherapy. Int J Mol Sci. 2019;20:2241.
28. Moufarrij S, Srivastava A, Gomez S, Hadley M, Palmer E, Austin PT, et al. Combining DNMT and HDAC6 inhibitors increases anti-tumor immune signaling and decreases tumor burden in ovarian cancer. Sci Rep. 2020;10:3470.
|
|
|||||||||||||||||||||||||||||||||||||||||||