AbstractPurposeThe prevalence of multi-morbidities with colorectal cancer (CRC) is known to be increasing. Particularly prognosis of CRC patients co-diagnosed with metabolic syndrome (MetSyn) was largely unknown. We aimed to examine the death risk of CRC patients according to the multiple MetSyn morbidities.
Materials and MethodsWe identified CRC patients with MetSyn from the electronic medical records (EMR) systems in five independent hospitals during 2006–2011. Information on deaths was jointly retrieved from EMR, cause of death registry and chronic disease surveillance as well as study-specific questionnaire. Cox proportional hazards regression was used to calculate the overall and CRC-specific hazards ratios (HR) comparing MetSyn CRC cohort with reference CRC cohort.
ResultsA total of 682 CRC patients in MetSyn CRC cohort were identified from 24 months before CRC diagnosis to 1 month after. During a median follow-up of 92 months, we totally observed 584 deaths from CRC, 245 being in MetSyn cohort and 339 in reference cohort. Overall, MetSyn CRC cohort had an elevated risk of CRC-specific mortality (HR, 1.49; 95% confidence interval [CI], 1.07 to 1.90) and overall mortality (HR, 1.43; 95% CI, 1.09 to 1.84) compared to reference cohort after multiple adjustment. Stratified analyses showed higher mortality risk among women (HR, 1.87; 95% CI, 1.04 to 2.27) and specific components of MetSyn. Notably, the number of MetSyn components was observed to be significantly related to CRC prognosis.
IntroductionThe metabolic syndrome (MetSyn) is characterized by a clustering of medical conditions related to cardiovascular diseases and diabetes mellitus type 2, including central obesity, hypertension, dyslipidemia, hyperglycemia and insulin resistance as well as other conditions such as proinflammatory state and prothrombotic state [1–3]. Studies indicate that prolonged chronic stress, sedentary lifestyle, lipodystrophy, and unbalanced diets may confer an increased risk of MetSyn [1,3]. Some of these factors were also reported to be related to the colorectal carcinogenesis [4].
Recently, there is cumulating evidence that MetSyn with its hormonal and systemic effects could increase the susceptibility to various cancers [5–11]. Epidemiological studies show association of MetSyn and/or its components with elevated risk of cancer including colorectal cancer (CRC) [2,6,9,12,13], which is ranked as the third most common neoplasms and the fourth most deadly malignancy in the world according to GLOBOCAN [14–16]. Biological mechanism underlying this association had been explained as a number of ways such as the growth hormone deregulation, cellular crosstalk, and vascular integrity factors [11,17–19].
To our knowledge, the effect of multi-morbidities of MetSyn on the prognosis of CRC patients has rarely been performed. In this study, we aimed to examine whether the mortality of CRC patients was impacted by co-diagnosis of MetSyn, with particular interests in exploring the single and collaborative effects of MetSyn components on death risk of CRC patients by sex. This will be able to aid in identifying individuals at risk and provide thoughts for clinical management and treatment.
Materials and Methods1. Study populationThis retrospective cohort study was conducted in five independent tertiary hospitals in China: Beijing Chao-Yang Hospital, Sichuan Cancer Institute/Hospital, the Affiliated Suzhou Science and Technology Town Hospital of Nanjing Medical University, the First Affiliated Hospital of Soochow University, and Harrison International Peace Hospital. We retrospectively reviewed the electronic medical records (EMR) of patients whose first diagnosis of cancer was CRC (International Classification of Diseases-10 [ICD-10] code C18-C20) from January 31, 2006 to December 31, 2011. All CRC cases were histologically confirmed and staging was assessed. Only participants aged between 35 and 79 was finally included. Patients co-diagnosed with any other cancers (ICD10: C00-C97 exclusive of C18-C20) and/or other severe diseases than MetSyn were excluded: (1) severe cardiovascular disease (acutemyocardial infarction [ICD10: I21], heart failure [ICD10: I50]) and severe cerebrovascular diseases (ICD10: I60-I69) for their weakness; (2) dementia and severe mental disorders for not being able to provide the accurate answers when questionnaire were administered for their impaired memory (Fig. 1).
2. MetSyn CRC cohortAny CRC patients with co-diagnosis of MetSyn from 24 months before CRC diagnosis to 1 month after CRC diagnosis (presurgery) were identified and included in MetSyn CRC cohort. Based on the International Diabetes Federation (IDF) criteria and the criteria from the American Heart Asociation, definition of MetSyn was adapted to our data as the presence of abodominal obesity, supplemented by at least two of the following: clinical diagnosis of hypercholesterolaemia, hypertension and diabetes mellitus type 2. Abodominal obesity (central obesity) was defined as waist circumstance: ≥ 90 cm for men and ≥ 80 cm for women around 2 years before CRC diagnosis.
3. Reference CRC cohort and propensity score matchingReference cohort was composed of CRC patients with no diagnosis of MetSyn. In order to mitigate the selection bias caused by co-diagnosis of MetSyn, multiple logistic regression analyses were used to control for the remaining imbalance due to the confounding difference between MetSyn CRC cohort and reference cohort by performing propensity score matching. Controls were first selected from the same hospital as the case was diagnosed during the same period for all five hospitals and further matched to the cases at a ratio of 2:1. Based on a multiple conditional logistic regression model including covariates age, sex, year at diagnosis, education achievement, stage at diagnosis, and site of CRC, the propensity score that was the probability of MetSyn of an individual conditional on a serial of observed demographic and clinical characteristics was calculated and we selected to use nearest neighbor propensity scores matching. Reference cohort was matched to cases with the minimum difference in propensity score.
4. Data collection and interview1) Sources of dataCauses of death have been accessed by using the following ways in combination: cause of death reporting system from Centre for Disease Control and Prevention (CDC) in each region; EMR system in hospitals; death certificate; maternal and child surveillance system; cancer registry data. Information of patients that was still unavailable by combining ways above was collected by phone interview and these patient comprised less than 5% of all.
In addition, demographic and clinical characteristics were collected by a combined retrieval of medical records and other above-mentioned data sources, supplemented by a survey using a structured questionnaire. The questionnaires were administered by trained interviewers. A pre-designed and validated questionnaire was used to collect information on related factors. Body weight at various stages were collected by questionnaire for time of diagnosis of CRC, 2 years before CRC diagnosis and 7 years before CRC diagnosis.
2) Study outcomesWe identified all CRC patients who died during January 2006 to December 2016 to make sure that each participants in the entire cohorts had at least five years of follow-up. Primary outcome was defined as death due to CRC (ICD-10 code C18-C20) and the secondary outcome was death from all the causes (ICD-10 code A00-Z99).
5. Statistical analysesThe Kaplan-Meier (K-M) model was used to calculate the probability of CRC-specific survival (CSS) and the probability of overall survival (OS) as a function of time. The differences between the K-M curves were tested for significance by the log-rank test. Cox PH regression was used to estimate hazard ratios (HR) and its 95% confidence interval (CI) for CRC mortality and overall mortality, controlling for potential confounding mainly from the selected demographic and clinical characteristics. Covariates adjusted in the model included: sex, age at diagnosis of CRC, year at diagnosis of CRC, highest education achievement, stage of CRC (I to IV), site of CRC, research centres. Both the MetSyn cohort and the reference cohort were followed from the date of CRC diagnosis until the earliest death, loss of follow-up, or the end of the study (December 31, 2016), whichever comes first. In addition, we used receiver operating characteristics (ROC) curve to predict the most dominant MetSyn morbidity in CRC patients, by comparing the higher area under the curves (AUC) for individual component and also different combinations of the MetSyn.
The K-M survival curves were drawn using the MedCalc software (MedCalc Software bvba, Ostend, Flanders, Belgium). All other analyses were performed using the SAS statistical package ver. 9.3 (SAS Institute Inc., Cary, NC). All p-values were based on two-sided tests, with the statistical significance level set to 0.05.
ResultsIn this study, MetSyn cohort composed of 682 CRC patients with co-diagnosis of MetSyn were identified from five tertiary hospitals during July 2006 through December 2011 and matched to the comparison cohort by propensity score matching at a ratio of 1:2. Table 1 shows the main demographic and clinical characteristics as well as treatment information for both MetSyn CRC cohort and reference CRC cohort. CRC were relatively more likely to be observed among men, those with no university education, and at the age of 60–69 as well as at the middle staging and at the site of colon. In general, patients in MetSyn cohort was proportionate to those in reference cohort according to categories of main covariates (p > 0.05). At the end of the follow-up, number of CRC patients who underwent radical resection were 559 (82%) and 1,146 (84%) among MetSyn and reference cohort, respectively. Patients with first diagnosis of diabetes, hypertension, hypercholesterolaemia, and overweight accounted for 23%, 30%, 26%, and 21% among MetSyn cohorts, respectively. Meanwhile, patients with 1, 2, and over 3 components of MetSyn were 437, 211, and 34.
After a mean of 7 years and 4,706 accumulated person-years of follow-up, we totally observed 402 deaths in the MetSyn cohort, among which 245 died of CRC diagnosis. Comparatively, there were a mean of 7.5 years and 10,094 person-years accumulated during the follow-up for the reference cohort. The overall mortality rate was 8.96 per 1,000 person-years in the MetSyn cohort compared to 6.10 per 1,000 person-years in the comparison cohort, and the CRC mortality rate was 5.47 per 1,000 person-years in the MetSyn cohort compared to 3.52 per 1,000 person-years in the comparison cohort (Table 2). Consequently, co-diagnosis with MetSyn for CRC patients was associated with an elevated HR of 1.49 (95% CI, 1.07 to 1.90) for CRC death and 1.43 (95% CI, 1.09 to 1.84) for overall death. K-M survival curves based on the MetSyn groups were performed for CRC survival (Fig. 2A) and OS (Fig. 2B).
The association was further stratified by age at diagnosis of CRC, sex, site of diagnosis, and staging of cancer. The increment of mortality risk was observed to be more prominent among women, cancer diagnosed in colon, and those in stage III and IV. Some of the insignificant results could be partially due to the reduced number of cases due to stratification.
As shown in Table 3, we accessed how the component/number of MetSyn multi-morbidities impact the mortality risk of CRC patients. Although the number of CRC patients reduced due to the stratified analyses, we still observed a series of significant mortality risk elevation. Sex played a markedly modifying effect on this observed association, that is, increment of CRC mortality for hypertension (HR, 1.63; 95% CI, 1.15 to 2.20) and hypercholesterolaemia (HR, 1.40; 95% CI, 1.19 to 1.88) in women versus diabetes (HR, 1.42; 95% CI, 1.01 to 2.25) in men with regard to CRC-specific mortality. The significant impact of overweight on the CRC mortality was not observed for both sexes in this study. Notably, we observed that number of MetSyn components was associated with mortality for both sex (p < 0.01): the more number of MetSyn components, the higher mortality risk from both CRC death and all causes of deaths.
The predictive ability for cause-specific survival and OS of CRC cases of MetSyn components and their combinations using ROC curves were assessed by comparing the difference of AUC. A moderate predictive ability were observed with AUCs ranging from above 55% to below 68%. As for the specific component of MetSyn, the highest AUC was observed for hypercholesterolaemia (64% for CRC death, 61% for overall death) and the lowest AUC was noted for overweight (55% for CRC death, 57% for overall death). Simultaneously, the relatively higher predictive ability of death risk occurred among CRC patients with any 2 combinations (63%–68%) than others.
DiscussionIn this multicentre retrospective cohort study, we totally identified 682 CRC patients with diagnosis of MetSyn and our findings demonstrated a 49% increment in CRC-specific mortality and a 43% increment in overall mortality comparing MetSyn CRC cohort with reference CRC cohort after propensity score matching. Further, elevation of mortality risk was observed to be more prominent among women, patients with cancer in the site of colon and those diagnosed at later stage. Although mortality risk of CRC patients varied by components of MetSyn and sex, we consistently observed that the more MetSyn components were co-diagnosed with CRC patients, the higher mortality risk was.
Our findings that death risk for CRC patients with multiple diagnoses of MetSyn components was always higher than those with single component showed that MetSyn should be treated as an integrated medical condition rather than its parts as for its impact on CRC prognosis. Observational studies found that male could be a risk factor for increased CRC risk. However, our study observed that female was associated with unfavorable CRC prognosis. More recently, MetSyn has been correlated with a number of cancers [9,12,20–22]. Epidemiological studies reported that there could be sex-based discrepancy for the association of MetSyn with common cancer such as CRC [10]. To our knowledge, the biological mechanisms underlying the female unfavorable prognosis among MetSyn CRC patients remain still unclear. Biological links between MetSyn and cancer risk had been involved as many as factors and signaling pathways described in deregulation of cytokine production, chronic inflammatory state, insulin-like growth factor system, and hormones and proinflammatory cytokines, among which hormone-related factors and signaling pathways were thought to be partially interpreted the high mortality risk observed among female CRC patients co-diagnosed with MetSyn [11].
The present study has a number of strengths. First, co-diagnosis of MetSyn with CRC during a 2-year period is relatively uncommon. Multicentre design with propensity score design increased sample size to acquire a better power and reduce the potential bias by better balancing the baseline characteristics between MetSyn CRC cohort and comparison CRC cohort. Second, definition of MetSyn was adapted to our study by combining several international criteria and the emphasis was put on the mechanism of colorectal carcinogenesis. Third, the follow-up for any study subjects was at least 5-year, which makes sure most of the mortality was observed during the study. In addition, multiple sources of outcome acquirement ensured the completeness of the data. However, limitations do exist for this study. One could be that lack of covariate information at individual level such as medications hindered the related analyses. However, the multiple matching could minimize the discrepancy from the major covariates. Additionally, central abodominal obesity is the necessary criteria to be included into the MetSyn cohort in our study and development of CRC could cause weight loss. While we did not find weight differences between cohorts at time of diagnosis of CRC, 2 years before CRC diagnosis and 7 years before CRC diagnosis (p > 0.05). Also, there is no internationally agreed definition for the MetSyn so far. In the present study, we adopted from both the IDF criteria and the criteria from the American Heart Asociation as a working definition to better service our study purpose. However there are more MetSyn components could be included according to other criteria such as the National Cholesterol Education Program Adult Treatment Panel III (NCEP-ATPIII), Blood and Lung Institute (AHA/NHBLI) or the WHO criterion [23]. The absence of other MetSyn components for example proinflammatory and prothrombotic states impeded to explore their impacts on CRC prognosis.
So far, there are a number of studies having examined the association of MetSyn with risk of CRC [2,9,12,13]. Claudio et al. [2] investigated the association of CRC risk with MetSyn components in Italy and Switzerland based on a multicentre case-control design. They found significant association between CRC risk and previous diagnosis of hypertension (OR, 1.24; 95% CI, 1.03 to 1.48) or hypercholesterolemia (OR, 1.26; 95% CI, 1.08 to 1.48) for men while significant association was not observed for women. Hypertension had been reported to be related to DNA repair synthesis and carcinogen uptake [24]. We could not find other previous studies specifically designed for investigating the biological mechanism of hypertension in CRC prognosis and plausible biological mechanism need to investigate in the future study. Recently, one study had indicated that hypercholesterolemia increased CRC incidence by reducing production of NKT and γδ T cells from hematopoietic stem cells, which may partially interpret the unfavorable survival of CRC patients with co-diagnosis of hypercholesterolaemia [25]. Association with prognosis was not performed in this study. Another study [13] aimed to assess the risk of CRC with type 2 diabetes based on a matched cohort design at a ratio of 1:1. Authors observed that type 2 diabetes associated with 1.3 times of CRC risk increment (HR, 1.26; 95% CI, 1.18 to 1.33). Longer duration of obesity was associated with CRC risk. In Me-Can (Metabolic Syndrome and Cancer) project [12], authors found increased CRC risk in men (HR, 1.25; 95% CI, 1.18 to 1.32) and in women (HR, 1.14; 95% CI, 1.06 to 1.22) with diagnosis of MetSyn after a mean follow-up of 12 years among 580,000 men and women. Furthermore, significant association was observed for body mass index (BMI; relative risk [RR], 1.07; 95% CI, 1.02 to 1.13), blood pressure (RR, 1.10; 95% CI, 1.02 to 1.18) and triglycerides (RR, 1.17; 95% CI, 1.06 to 1.28) in men as well as for BMI (RR, 1.08; 95% CI, 1.01 to 1.15) in women. Combination of metabolic factors was related to a higher risk of CRC.
Yet, there were few data on the association of prognosis of CRC and MetSyn so far. A hospital-based retrospective cohort conducted in Kaiser Permanente in United States [26] had investigated the impact of MetSyn and obesity on survival among CRC patients diagnosed at early stage. They found that obese patients with MetSyn had been related to both a worse OS (HR, 1.45; 95% CI, 1.12 to 1.82) and a worse CSS (HR, 1.49; 95% CI, 1.09 to 2.02) compared to non-obese patients without MetSyn. Obesity or MetSyn alone, however, did not decrease survival rate. A study conducted in China [27] which aimed to investigate the effect of preoperative MetSyn on CRC mortality for patients after a radical resection observed a significant shorter survival time for CRC patients with MetSyn compared to those with no MetSyn (50.9 months vs. 170.3 months, p < 0.01) after a median follow-up of 58.6 months, particularly for patients with diagnosis of hyperglycemia (44.4 months vs. 170.3 months, p < 0.01). The magnitude of the association was more prominent for CRC patients diagnosed at stage I/II (HR, 3.94; 95% CI, 2.65 to 5.85) and invasion depth T1/T2 (HR, 5.41; 95% CI, 2.54 to 11.50).
Although the elevated CRC risk was more prominent among men than women, increased mortality risk for CRC patients was more eminent among women in our study. The unfavorable prognosis noted among colon cancer, not rectum, may reflect the later diagnosis of colon cancer than rectum cancer since the clinical symptom of rectum cancer usually urge the patients to look for clinical counselling. Inconsistent results between studies could be caused by a number of reasons. First, variation of MetSyn definitions could hinder the direct comparability of the results to some extents, whereas the impacts of specific MetSyn component on the CRC mortality were basically consistent in different studies. We, here, combined the definition from both IDF criteria [3] and ATPIII from The American Heart Association [1] to better adapt the data to the shared metabolic basis of MetSyn and CRC proposed. Second, hospital-based cohort study was the most commonly used design by most studies as for the prognosis study, in which the robustness of the results could be impacted by the duration of follow-up and staging of cancer diagnosis.
To sum up, this is one of the first studies examining whether the CRC prognosis will be impacted by co-diagnosis of MetSyn, wherein our results suggest that MetSyn should be better treated as an integrated medical condition with its components collaboratively as for the prognosis management and therapy for CRC. Effect of specific MetSyn components on CRC patient survival differs by sex.
NotesEthical Statement Our research protocals were approved by the institutional review boards of Soochow University and the respective ethical committees at the participating hospitals (Q413900215). The implement of the current study adhered to the tenets of the Declaration of Helsinki of the World Medical Association with regard to scientific research on human subjects. The written informed consent forms for participation and publication were obtained for all study participants. In order to preserve anonymity, individual record of all participants was de-identified before the analysis. Author Contributions Conceived and designed the analysis: Feng Q, Shu X. Collected the data: Feng Q, Xu L, Li L. Contributed data or analysis tools: Qiu J, Huang Z, Jiang Y, Wen T, Lu S, Meng F. Performed the analysis: Feng Q, Xu L, Li L, Meng F, Shu X. Wrote the paper: Feng Q, Xu L, Shu X. Financial support: Shu X. AcknowledgmentsThe corresponding author would like to thank all the co-workers for collecting, managing and maintaining the data used in this analysis. We also appreciate Soochow University for providing the financial support to conduct the present study. This work was jointly supported by Scientific Research Foundation for Talented Scholars in Soochow University, China, The Nuclear Energy Development Project, China (no. 2016-1295), and a Project of the Priority Academic Program Development of Jiangsu Higher Education Institutions China.
Fig. 1Flow diagram of study participants. CRC, colorectal cancer; MetSyn, metabolic syndrome; PSM, propensity score matching. 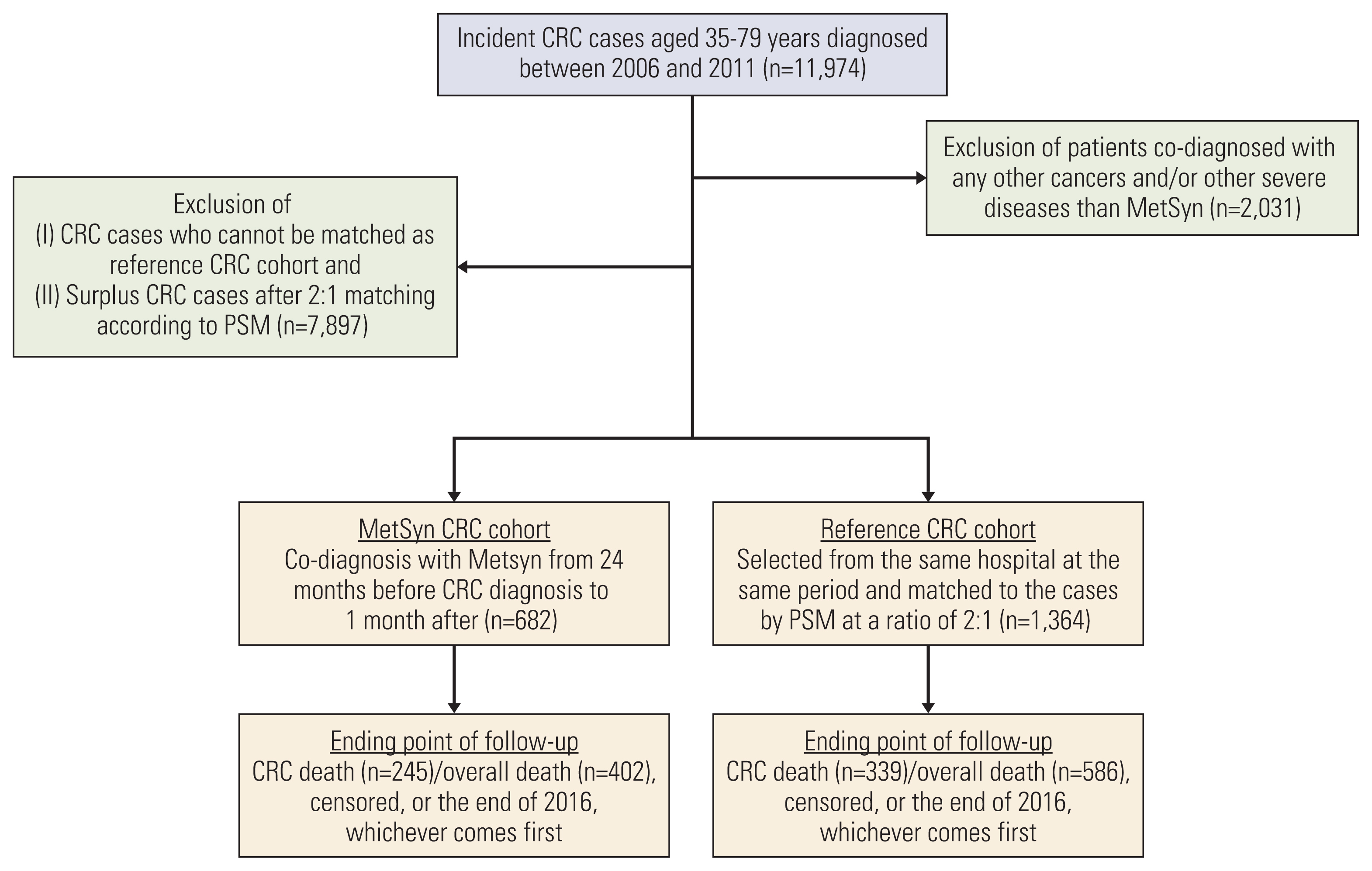
Fig. 2(A) Probability of cause-specific survival of colorectal cancer (CRC) patients co-diagnosed with metabolic syndrome (MetSyn) in comparison with CRC patients without MetSyn diagnosis. (B) Probability of overall survival of CRC patients co-diagnosed with MetSyn in comparison with CRC patients without MetSyn diagnosis. 
Table 1Characteristics of MetSyn CRC cohort and reference CRC cohort by propensity score matched controls Table 2Association between co-diagnosis of MetSyn and the risk of CRC-specific and overall mortality by demographic and clinical characteristics Table 3Association between co-diagnosis of MetSyn and the risk of CRC-specific and overall mortality by component and number of components of MetSyn References1. Grundy SM, Brewer HB Jr, Cleeman JI, Smith SC Jr, Lenfant C, et al. American Heart AssociationDefinition of metabolic syndrome: report of the National Heart, Lung, and Blood Institute/American Heart Association conference on scientific issues related to definition. Circulation. 2004;109:433–8.
2. Pelucchi C, Negri E, Talamini R, Levi F, Giacosa A, Crispo A, et al. Metabolic syndrome is associated with colorectal cancer in men. Eur J Cancer. 2010;46:1866–72.
3. Alberti KG, Zimmet P, Shaw J. IDF Epidemiology Task Force Consensus GroupThe metabolic syndrome: a new worldwide definition. Lancet. 2005;366:1059–62.
5. Esposito K, Chiodini P, Colao A, Lenzi A, Giugliano D. Metabolic syndrome and risk of cancer: a systematic review and meta-analysis. Diabetes Care. 2012;35:2402–11.
6. Ishino K, Mutoh M, Totsuka Y, Nakagama H. Metabolic syndrome: a novel high-risk state for colorectal cancer. Cancer Lett. 2013;334:56–61.
7. Stocks T, Van Hemelrijck M, Manjer J, Bjorge T, Ulmer H, Hallmans G, et al. Blood pressure and risk of cancer incidence and mortality in the Metabolic Syndrome and Cancer Project. Hypertension. 2012;59:802–10.
8. Ulmer H, Bjorge T, Concin H, Lukanova A, Manjer J, Hallmans G, et al. Metabolic risk factors and cervical cancer in the metabolic syndrome and cancer project (Me-Can). Gynecol Oncol. 2012;125:330–5.
9. Kabat GC, Kim MY, Peters U, Stefanick M, Hou L, Wactawski-Wende J, et al. A longitudinal study of the metabolic syndrome and risk of colorectal cancer in postmenopausal women. Eur J Cancer Prev. 2012;21:326–32.
10. Esposito K, Capuano A, Giugliano D. Metabolic syndrome and cancer: holistic or reductionist? Endocrine. 2014;45:362–4.
11. Mendonca FM, de Sousa FR, Barbosa AL, Martins SC, Araujo RL, Soares R, et al. Metabolic syndrome and risk of cancer: which link? Metabolism. 2015;64:182–9.
12. Stocks T, Lukanova A, Bjorge T, Ulmer H, Manjer J, Almquist M, et al. Metabolic factors and the risk of colorectal cancer in 580,000 men and women in the metabolic syndrome and cancer project (Me-Can). Cancer. 2011;117:2398–407.
13. Peeters PJ, Bazelier MT, Leufkens HG, de Vries F, De Bruin ML. The risk of colorectal cancer in patients with type 2 diabetes: associations with treatment stage and obesity. Diabetes Care. 2015;38:495–502.
14. Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–86.
15. Center MM, Jemal A, Smith RA, Ward E. Worldwide variations in colorectal cancer. CA Cancer J Clin. 2009;59:366–78.
16. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
17. Saetang J, Sangkhathat S. Diets link metabolic syndrome and colorectal cancer development (Review). Oncol Rep. 2017;37:1312–20.
18. Vargas T, Moreno-Rubio J, Herranz J, Cejas P, Molina S, Gonzalez-Vallinas M, et al. Genes associated with metabolic syndrome predict disease-free survival in stage II colorectal cancer patients: a novel link between metabolic dysregulation and colorectal cancer. Mol Oncol. 2014;8:1469–81.
19. Braun S, Bitton-Worms K, LeRoith D. The link between the metabolic syndrome and cancer. Int J Biol Sci. 2011;7:1003–15.
20. Russo A, Autelitano M, Bisanti L. Metabolic syndrome and cancer risk. Eur J Cancer. 2008;44:293–7.
21. Marrero JA, Fontana RJ, Fu S, Conjeevaram HS, Su GL, Lok AS. Alcohol, tobacco and obesity are synergistic risk factors for hepatocellular carcinoma. J Hepatol. 2005;42:218–24.
22. Xue F, Michels KB. Diabetes, metabolic syndrome, and breast cancer: a review of the current evidence. Am J Clin Nutr. 2007;86:s823–35.
23. Alberti KG, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med. 1998;15:539–53.
24. Norden A, Schersten B, Thulin T, Pero RW, Bryngelsson C, Mitelman F. Letter: hypertension related to D.N.A. repair synthesis and carcinogen uptake. Lancet. 1975;2:1094.
25. Tie G, Yan J, Khair L, Messina JA, Deng A, Kang J, et al. Hypercholesterolemia increases colorectal cancer incidence by reducing production of NKT and gammadelta T cells from hematopoietic stem cells. Cancer Res. 2017;77:2351–62.
|
|
||||||||||||||||||||||||||||||||||||||||