INTRODUCTION
Taxanes are important anti-tumor drugs endowed with a unique mechanism of action: they inhibit microtubule (MT) disassembly (1,2). The taxanes, paclitaxel and docetaxel, are relatively novel anti-mitotic agents that are under extensive investigation in clinical trials (3,4). Both taxanes have demonstrated significant activity against many solid tumors as a either a single agent or in combination with other chemotherapeutic agents (5,6). The taxanes in current clinical use are effective against a broad spectrum of human tumors, in particular, ovarian and breast carcinomas.
Paclitaxel, a potent drug of natural origin isolated from the bark of the Pacific yew, Taxus brevifolia (7), is currently used in the treatment of ovarian, lung, and breast cancer. Initial studies on the mechanism of action of paclitaxel have demonstrated that this drug alters MT assembly by inhibiting MT depolymerization and changing MT dynamics. These effects result in the disruption of the normal reorganization of the MT network required for mitosis and cell proliferation. Paclitaxel-treated cells are unable to proceed normally through the cell cycle and are arrested in the G2/M phase (8). Although treatment of various tumor cells with paclitaxel in-vivo and in-vitro induces apoptosis and paclitaxel has been shown to regulate the expression of several apoptosis-related proteins, including bcl-2 (9~11), bax (10), bcl-X (11), p21waf (12), and tumor necrosis factor (TNF)-α (13,14) the exact mechanism by which this drug induces apoptosis is not yet known.
This report describes the first evaluation using 2-DE/MALDI-TOF-MS to profile differential protein expression and to analyze antineoplastic mechanism-related proteins in HeLa cervical carcinoma cells during paclitaxel-induced cell death.
MATERIALS AND METHODS
1) Cell culture and cytotoxicity assay
HPV-18 positive HeLa cervical carcinoma cells were grown in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal bovine serum at 37℃ in 5% CO2. The cytotoxic studies were carried out using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazonium bromide (MTT) (Sigma, St. Louis, MO) representing the percentage of growth inhibition induced by increasing drug concentrations. The HeLa cells (1×104/well) were incubated with paclitaxel for 12 h, 24 h, 36 h, 48 h and 72 h on the 96 well plates. After a 4 h incubation with MTT substrate (20 mg/ml), the culture medium was removed and DMSO added. The cytotoxicity was evaluated with reference to the IC50 value. The tests were performed at least three times.
2) Two-dimensional gel electrophoresis and image analysis
HeLa cells were treated with 0.01 µM paclitaxel and harvested by trypsinization after 24 h incubation. Cell pellets were solved in lysis buffer containing 7 M urea, 2 M thiourea and 4% CAHPS. After sonication, 0.05 mg of total protein was loaded onto an immobilized pH 3~10 (linear) IPG strip (Amersham Bioscience, Arlington Heights, IL) at 20℃ using a Ettan IPG phor (Amersham Bioscience). The IPG strips were rehydrated overnight in a solution of 7 M urea, 2 M thiourea, 4% CHAPS, 45 mM DTT, 0.5% IPG buffer, 400 mM Tris and a trace of bromophenol blue prior to use. IEF was carried out using the following conditions; 100 V, 50 VH; 300 V, 150 VH; 600 V, 300 VH; 1,000 V, 500 VH; 3,000 V, 1,500 VH; 6,000 V, 36,000 VH. Focused gels were stored at -20℃ prior to SDS-PAGE, then incubated for 15 min in equilibration solution (50 mM Tris-Cl, 6 M urea, 30% glycerol, 2% SDS, 0.002% BPB) with 100 mg/ml DTT, followed by 15 min in equilibration buffer with 25 mg/ml iodoacetamide and rinsed with SDS-PAGE buffer (25 mM Tris-Cl, 192 mM glycine, 0.1% SDS). Proteins were visualized by silver stain.
The stained 2D gels were scanned with a Bio-Rad Scanner (Bio-Rad, Philadelphia, PA). The image analysis and 2D gel proteome database management were done using the PDQuest software 6.2.1 (Bio-Rad). For identification of proteins by mass spectrometry, matching was done between analytical silver-stained gels and preparative gels in order to correlate the precise position of the spots to be excised.
3) MALDI-TOF-MS
In-gel digestion of protein spots on silver stained gels was performed. After the completion of staining, the gel slab was washed twice with water for 10 min. The spots of interest were excised with a scalpel, cut into pieces and put into 1.5 ml microtubes. Enough 25 mM Ammonium bicarbonate buffer, pH 8.0/50% acetonitrile (1:1) (Sigma) was added to the tube to cover the gel pieces, and the tube was incubated at room temperature for 15 min. The liquid was discarded and washing was repeated. After the pieces were soaked by dehydration in 100% acetonitrile, which was then removed, they were dried in a Speed-Vac. The dried gel pieces were rehydrated with 15 µl of trypsin solution (10 µg/ml in 25 mM ammonium bicarbonate buffer, pH 8.0) and incubated at 37℃ for 12~14 h. Tryptic peptides were extracted using 50% Acetonitrile/5% TFA. After removal of acetonitrile by centrifugation in a Speed-Vac, the peptides were concentrated by using C18Zip-Tip (Millipore Corp., Bedford, MA) and eluted with 2 µl of 100% acetonitrile and directly spotted on the sample plate of a MALDI-TOF-MS. Finally α-cyano-4-hydroxycinnamic acid (0.5 µl of 10 mg/ml) was applied to each spot, and the spots were air-dried at room temperature prior to acquiring mass spectra. Peptide mass profiles produced by MALDI-TOF were analyzed using MS-FIT and Mascot. Peptide masses were compared with the theoretical masses derived from the sequences contained in SWISS-PROT and NCBI data banks.
4) Western-blot
Samples were mixed with Laemmli's loading buffer, boiled for 5 min, and subjected to 12% SDS-PAGE at 130 V followed by electroblotting to nitrocellulose membrane for 2 h at 80 V. Membranes were blocked for 1 h with 5% skim milk in TBS at room temperature and subsequently probed overnight with anti-TRAIL (Santa Cruz Biotechnology, Santa Cruz, CA), anti-caspase-8 (Santa Cruz Biotechnology), anti-c-myc (Santa Cruz Biotechnology), anti-caspase-5 (Santa Cruz Biotechnology), anti-bcl-2 (Santa Cruz Biotechnology) and anti-β-actin (Sigma). The membranes were rinsed and incubated with a HRP-conjugated secondary antibody. Following the secondary antibody incubation, the membranes were rinsed and bound antibodies were detected using enhanced chemiluminescence according to the manufacturer's instructions.
RESULTS
1) Cytotoxic effects by paclitaxel treatment in cervical carcinoma cells
The cytotoxic effects of paclitaxel were determined in HeLa cervical carcinoma cells. Following in vitro treatment of the cells with increasing concentrations of paclitaxel for 12 h, 24 h, 36 h, 48 h and 72 h, cell proliferation was evaluated using the MTT assay and dose- and time-response curves were plotted. To measure the IC50, the concentration inhibiting growth by 50%, cultured HeLa cells were treated with paclitaxel at six different concentrations (0, 0.01, 0.02, 0.04, 0.06, 0.08, and 0.1 µM). The results showed that the cell survival rate was reduced significantly by paclitaxel treatment in a dose- and time-dependent manner (Fig. 1).
2) Proteomic analysis to identify changes in protein expression stimulated by paclitaxel treatment in cervical carcinoma cells
To analyze the underlying mechanisms and identify down-stream mediators that are unique to the effects of paclitaxel, we performed a proteomic analysis to identify target-specific proteins important for enhanced cell death. An expression profile map from control-treated cells was used to generate a master expression profile map. This master pattern of protein expression was used to identify differences after treatment with paclitaxel. Three pairs of gels from different batches of control and paclitaxel-treated HeLa cells were analyzed for quantitative spot comparisons with the image analysis software. Treatment-dependent differences in expression are highlighted with arrows (Fig. 2). The goal was to identify and functionally characterize crucial proteins that lie upstream of the actual event of drug-induced apoptosis. To identify differentially expressed proteins in HeLa cells, we chose 47 proteins demonstrating significant changes in paclitaxel-treated cells compared to control cells. These proteins were identified by MALDI-TOF-MS and database searches with high confidence based upon high scores and sequence coverage. In this study, 24 proteins were up-regulated and 23 proteins were down-regulated by paclitaxel treatment as determined by spot volume (p<0.05). The identified proteins were functionally classified into 14 groups: apoptosis, immune response, cell cycle, metabolism, chaperone, protein transportation, DNA repair, replication, transcriptional regulation, translational regulation, ion channel, growth factor/oncogene, signal transduction, and structural organization/cell motility.
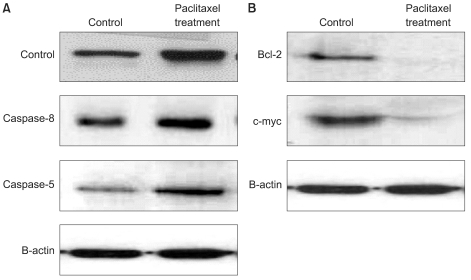
3) Anti-proliferative and anti-cancer effects of paclitaxel treatment
Western blots were performed for apoptosis- and transformation-related proteins to confirm expression patterns derived from proteome analysis after treatment of paclitaxel; tumor necrosis factor ligand superfamily member (TRAIL), caspase-8, caspase-5, bcl-2 and c-myc (Fig. 3). Our experiments showed paclitaxel engaged the membrane DR-mediated apoptotic pathway involving activation of caspase-8 in a TRAIL-dependent fashion and the mitochondrial apoptotic pathway involving down-regulation of bcl-2 by cytosolic cytochrome c release. Also, we found paclitaxel treatment induced down-regulation of myc proto-oncogene (c-myc). Expression data obtained with 2-DE proteomics were strongly correlated with the results of the Western blot.
DISCUSSION
Paclitaxel, one of the best anti-cancer drugs found in nature over the past several decades, has been approved by the United States FDA for chemotherapy of a wide spectrum of cancers, including breast cancer, ovarian cancer, small/non-small cell lung cancer, and AIDS related Kaposi's sarcoma (15,16). In addition to its excellent anti-tumor and anti-proliferative activities, paclitaxel exerts potent and sustained inhibitory effects on cancer cell proliferation and migration in cell culture. Owing to binding with tubulin molecules, paclitaxel could enhance the polymerization or suppress the dynamic instability of microtubules (17,18), affecting the assembly and disassembly of cytoskeletons and thus leading to the cell cycle blockage in G2/M (19~21).
We performed proteomic analysis to find the altered proteins participated in paclitaxel-induced anti-proliferative and antineoplastic effects in cervical cancer cells. Paclitaxel treatment mainly elevated apoptosis (caspase-8, -5 and TRAIL protein, etc.), immune response (lymphocyte cytosolic protein 2 and protein-tyrosine phosphatatase, etc.), and cell cycle check point (cyclin H, mitotic checkpoint BUB3 and BTG3 protein, etc.), related proteins. On the other hand, paclitaxel treatment diminished growth factor/oncogene (c-myc and src substrate cortactin, etc.), DNA replication (eukaryotic initiation factor 4 and Ruv Blike 2, etc.), and transcription regulation (activated RNA pol II transcriptional coactivator p15 and zinc finger protein 265, etc.) related proteins.
Also, we could suggest that dual apoptotic pathways by paclitaxel treatment in HeLa cervical cancer cells may exist due to 1) membrane DR-mediated apoptotic pathway: membrane DR (TRAIL cell surface death receptor) → caspase-8 activation → caspase-3 activation → apoptosis, 2) cytochrome c-dependent apoptotic pathway: cytochrome c release → bcl-2 down-regulation → caspase-9 activation → caspase-3 activation → apoptosis.
Chemotherapy-induced changes in cervical cancer cells may be caused by proteins that are secreted by damaged tumor cells (22,23). It has been shown that exposure of cervical cancer cells to paclitaxel induces a change in the protein profile of these cells in vitro. Alterations in the protein constituents after paclitaxel treatment also may reflect a cellular response to a toxic agent (24,25). Therefore, proteome changes potentially may be used as early markers of tumor response or may herald or even mediate paclitaxel-induced, systemic side effects. The development of proteomics-based techniques offers excellent new opportunities for analysis of drug targets in human cancers.
CONCLUSIONS
The proteome profiling technique provided a broad-base and effective approach for the identification of protein changes induced by paclitaxel and showed anti-proliferative activity through the membrane death receptor-mediated apoptotic pathway and the mitochondrial-mediated pathway by paclitaxel treatment. Proteomic technology will contribute to a better understanding of the mechanisms of action of this antineoplastic drug at the molecular level and to the future development of novel therapeutic drug treatments based upon current therapies.